Monitoring of the internal environment in intensive care
Glycemia[edit | edit source]
Urea and creatinine[edit | edit source]
- Indicators of renal function;
- when urea rises but creatinine does not, it is an increased catabolism;
- worsening of renal function without increasing creatininemia may be bleeding into large muscle groups or into the GIT.
Total protein and albumin[edit | edit source]
- Albumin - oncotic pressure, pH!;
- protein losses are sometimes compensated by alternative colloidal solutions (dextran, gelatin…), because the administered protein can be deposited somewhere outside the bloodstream (eg into the lungs…).
Ions[edit | edit source]
Osmolality[edit | edit source]
Osmolality is the amount of osmotically active substances dissolved in a unit mass of solvent. It is usually expressed in Osm/kg or in fractions of this unit.
Osmolality is equal to the product of molality and the number of particles formed by the dissociation of one molecule.
However, most body fluids are real, i.e., non-ideal solutions where the real osmolality is less than the ideal osmolality. To obtain the value of the real osmolality, you need to know the osmotic coefficient of the solution, by which the value of the ideal osmolality of the solution is multiplied.
The physiological value of plasma osmolality is about 300 mOsm/kg of water.[1].
See Internal environment for more details .
Blood gases[edit | edit source]
- acid-base balance, oxygen parameters
Lactate[edit | edit source]
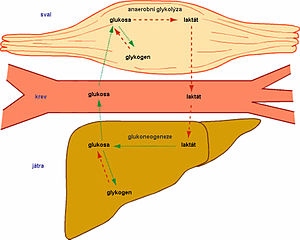
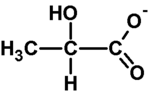
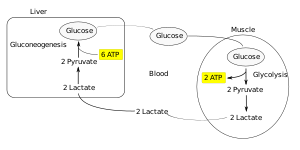
Lactate is a conjugate base of lactic acid, which is formed during anaerobic glycolysis by the conversion of pyruvate by lactate dehydrogenase (LD). The level of lactate in the blood is determined by the ratio between its production and its breakdown (gluconeogenesis) in the liver. Hyperlactataemia, and later lactic acidosis, arises either from overproduction or from insufficient utilization of lactate. Lactate is involved (in descending order): skin, erythrocytes, brain, muscles, intestinal mucosa, leukocytes, platelets. Lactate is further transported by the blood to the liver, where it is used for gluconeogenesis (Cori cycle), a smaller part of lactate is used by the kidneys (for gluconeogenesis and urinary excretion), the rest is metabolized by the myocardium and other organs. The accumulation of lactate in the muscles causes a decrease of the pH, and this causes muscle fatigue and pain.
References limits: lactate in blood serum = 0.6–2.1 mmol/l [2].
The concentration of lactate in the blood changes rapidly after collection - its concentration increases due to glycolysis in blood elements
Hyperlactacidemia[edit | edit source]
Causes:
- tissue hypoxia
- ischemia and tissue hypoperfusion
- hypoventilation (spontaneous or controlled)
- intensive short-term sports activities
- poisonings, that lead to tissue hypoxia (e.g. CO poisoning, methemoglobinemia, cyanide poisoning)
- lactate overproduction during ethanol intoxication and some metabolic defects (mitochondrial disorders, glycogenosis type I, etc.)
- insufficient lactate breakdown in hepatopathy
The role of lactate in glycolysis[edit | edit source]
In the absence of oxygen (absolute and relative), phosphoenolpyruvate is hydrolyzed to pyruvate during glycolysis to release energy. Pyruvate is further reduced (hydrogenated) to lactate using lactate dehydrogenase (LD). The hydrogen donor is NADH+H + , which is oxidized to NAD + in this reaction . The NAD + level needs to be maintained for anaerobic glycolysis. [4]
Although the reaction of converting pyruvate to lactate is reversible, LD prefers the formation of lactate rather than its oxidation. At the same time, lactate is not further metabolized by another mechanism. If the reaction is to be reversed, pyruvate must be rapidly removed.
Lactate breakdown[edit | edit source]
Cori cycle . Lactate is transported by blood to the liver, where it is re-oxidized to pyruvate. Pyruvate is converted back to glucose in the liver for energy consumption.
See the Gluconeogenesis page for more detailed information .
Clinical notes[edit | edit source]
- Values for shock :
- 1.3–4.4 mortality 18–22%;
- 4.5-8.9 - 73%;
- above 13 – 100%;
- an increase in lactate in sepsis is a sign of the transition to a metabolically decompensated stage. [2]
Lactate degradation[edit | edit source]
Lactate is transported by the blood to the liver, where it is back-oxidized to pyruvate. Pyruvate is converted back to glucose in the liver with energy consumption.
Acute phase reactants[edit | edit source]
The acute phase reaction is a physiological process that manifests itself in the systemic release of inflammatory mediators, due to the development of pathological processes (inflammation, trauma, surgery, myocardial infarction, childbirth, tumor processes, stress, excessive physical activity).
Mediators serve to ensure the overall response of the organism, communication and regulation of ongoing events. It also forms general symptoms (fever, fatigue, malaise, muscle and joint pain). Substances, whose synthesis arises as a result of known pathology or when their concentration corresponds to the degree of tissue damage, are of clinical importance. We call such substances markers (by determining them we can confirm or exclude the diagnosis of another disease.
Significance of acute phase positive reactants
The set of acute phase proteins is quite diverse. Nevertheless, depending on the effect, most of them can be classified into one of the following groups:
Components of the immune response
Some acute phase reactants are directly involved in the elimination of noxious substances, which has caused inflammation. Other proteins have a role in removing damaged cells or modulating the immune response. This includes e.g.
- C-reactive protein,
- complement components, in particular C3 and C4,
- tumor necrosis factor α (TNF-α), interleukin 1 (IL-1) and interleukin 6 (IL-6)
Protection against collateral tissue damage
During the acute phase, substances are mainly released from phagocytes and disintegrating cells to destroy the noxious substance, that caused the inflammation and to "dissolve" the damaged tissue. They are mainly proteolytic enzymes and reactive oxygen species. The effect of these substances should be limited, so that they act only where they have - i.e. so that the collateral tissue damage is as small as possible. Therefore, we find among the reactants of the acute phase.
Protease inhibitors
Proteins that reduce the production and availability of reactive oxygen species
These are not only scavengers of reactive oxygen species in the true sense of the word, but also proteins that bind and stabilize transition metals and their complexes. This reduces the formation of ROS in the Fenton reaction and similar processes. These include:
- haptoglobin,
- hemopexin,
- ferritin,
- ceruloplasmin.
Transport of waste products generated during inflammation
In addition to the hemoglobin and hemopexin mentioned above, this probably includes serum amyloid A (SAA)
Coagulation factors and proteins involved in tissue regeneration
The significance of some acute phase positive reactants remains unknown, although they may be clinically important proteins (used as inflammatory parameters). For example, procalcitonin (PCT).
Rate of change of acute phase reactant concentration
The plasma concentrations of the different acute phase reactants change at different rates. According to the time since the onset of the disease, during which it changes, we divide the acute phase reactants into three groups:
- Early acute phase proteins
- Acute phase proteins with a mean response time
- Late acute phase proteins
Early acute phase proteins[edit | edit source]
Early acute phase proteins are proteins with a very short biological half-life. Changes in their plasma concentrations are evident as early as 6-10 hours after the onset of the disease. The rise usually peaks during the second and third days. The main representatives are mainly C-reactive protein (CRP) and serum amyloid A (SAA). More recently, procalcitonin (PCT) is used in clinical practice.
C-reactive protein
C-reactive protein (CRP) is one of the most important acute phase reactants. It is a protein that plays the role of opsonin. It got its name due to the fact that it precipitates with the so-called C-polysaccharide of pneumococci [5]. It is part of innate immunity .
The plasma concentration of CRP increases already 4 hours after the induction of the acute phase reaction and during the first two days its concentration increases more than 100 times. The maximum concentration is reached in 24-48 hours, the half-life of CRP is approximately 24 hours. [6]
Physiological plasma concentration is up to 8-10 mg/l [7]. The rapid and high rise in CRP (typically to values above 50 mg/l) is accompanied mainly by acute bacterial infections, less usually also fungal infections. Viral infections, on the other hand, are characterized by a relatively small increase in CRP (usually below 25 mg/l)[8] . Therefore, determining the plasma concentration of CRP helps to decide whether to start antibiotic treatment. [5] Successful antibiotic therapy results in a rapid decrease in CRP, while an increase persists with unsuccessful treatment.
By determining CRP, the risk of postoperative infection can be detected. On the third day after the operation, its concentration should drop rapidly to normal. A persistent increase or only a partial decrease, followed by a further increase, indicates the presence of infection or other inflammatory complications.
A slight increase in CRP is also accompanied by myocardial infarction. In general, mildly elevated CRP levels (usually around 10 mg/l) are among the signs of high cardiovascular risk. [9]Monitoring CRP levels is also useful in monitoring autoimmune diseases.[10]
The disadvantage of CRP is its low specificity. Unlike procalcitonin, it does not indicate the severity of the organ disorder, but only the presence of the infection. These two markers do not replace each other, but complement each other.
Structure[edit | edit source]
The CRP molecule is made up of 5 non-covalently linked units , each of which is made up of a single non-glycosylated polypeptide chain. CRP belongs to the group of petraxins.
Function[edit | edit source]
The main functions of CRP include:
- Opsonization, the principle of which consists in the binding of CRP to phosphatidylcholine, which is part of bacterial walls.
- Complement activation,
- stimulation of phagocytic cells,
- the ability to bind the C-polysaccharide of the Streptococcus pnemoniae cell wall.
Procalcitonin
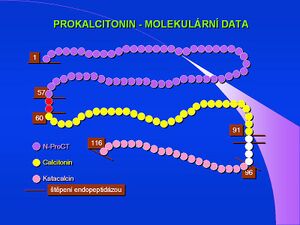
In recent years, procalcitonin (PCT) has been used as an acute phase reactant in research and clinical practice. This 116 amino acid protein, with a molecular weight of 13,000, is physiologically produced by thyroid C-cells as a precursor of the hormone calcitonin. However, especially in generalized bacterial infections, other cells, mainly neuroendocrine cells of the lung and intestine, but also cells of parenchymal organs and in sepsis practically all tissues and cell types begin to produce it. [11] The concentration of this protein in the plasma then rises sharply. PCT released during sepsis is not converted to calcitonin.[12]The exact physiological significance of procalcitonin is far from clear; it is thought to be involved in the regulation of inflammation and to have analgesic effects. The half-life of procalcitonin is 1 day, and after immune stimulation, its serum concentration increases about twenty-fold within 2-3 hours. The increase can be observed only in generalized bacterial, fungal and protozoal infections, it does not occur in viral infections. Less significant increases can be found in polytraumas, burns and after extensive abdominal operations.
See the Procalcitonin page for more detailed information .
Acute phase proteins with a mean response time[edit | edit source]
Acute phase proteins with a medium response time are proteins whose concentrations change 12–36 hours after the onset of the disease and the maximum is reached at the end of the first week. These include α1-acid glycoprotein (orosomucoid), α1-antitrypsin, haptoglobin and fibrinogen.
Late acute phase proteins[edit | edit source]
Late acute phase proteins are represented by complement components C3 and C4 and ceruloplasmin, which do not develop changes until 48-72 hours after the onset of the disease. The increase in concentrations is less pronounced in comparison with the two previous groups of proteins and peaks only after 6-7 days.
Acute phase negative reactants[edit | edit source]
Acute phase negative reactants are proteins whose levels decrease during acute exercise. The main representatives are albumin, prealbumin and transferrin. They are less important than positive reactants for monitoring and evaluating the course of the load response. However, they are often used as a criterion for protein synthesis in the liver and as indicators of malnutrition.
Liver function tests[edit | edit source]
- Bilirubin, AST, ALT, GGT;
- However, we encounter pathological findings in almost all patients at ICU - they are mostly hypovolemic (hypoxia), have an infection, due to drugs and parenteral nutrition.
More detailed information can be found on the Liver tests page .
Urine examination[edit | edit source]
- diuresis - performed daily
Links[edit | edit source]
References[edit | edit source]
- SCHNEIDERKA, Petr, et al. Kapitoly z klinické biochemie. 2. edition. Prague : Karolinum, 2004. ISBN 80-246-0678-X.
- ↑ ŠVÍGLEROVÁ, Jitka. Osmolalita [online]. The last revision 2009-02-18, [cit. 2010-11-12]. <https://web.archive.org/web/20160416224559/http://wiki.lfp-studium.cz/index.php/Osmolalita>.
- ↑ a b SCHNEIDERKA, Petr, et al. Kapitoly z klinické biochemie. 2. edition. Prague : Karolinum, 2004. ISBN 80-246-0678-X.
- ↑ https://nemocnicepodhorska.agel.cz/obsah/oddeleni/okbh_bruntal/_downloads/lab/laktat.pdf
- ↑ LEDVINA, Miroslav – STOKLASOVÁ, Alena – CERMAN, Jaroslav. Biochemie pro studující medicíny I. díl. 1. edition. Prague : Karolinum, 2005. 118; 274 pp. ISBN 80-246-0849-9.
- ↑ a b RACEK, Jaroslav, et al. Klinická biochemie. 2. edition. Prague : Galén, 2006. 329; 71 pp. ISBN 80-7262-324-9.
- ↑ ZIMA, Tomáš. Velký lékařský slovník online : Normální hodnoty [online]. [cit. 2020-02-13]. <http://lekarske.slovniky.cz/normalni-hodnoty>.
- ↑ ZIMA, Tomáš. Velký lékařský slovník online : Normální hodnoty [online]. [cit. 2020-02-13]. <http://lekarske.slovniky.cz/normalni-hodnoty>.
- ↑ KESSLER, Siegfried. Laboratorní diagnostika. 1. edition. Prague : Scientia medica, 1993. 252 Memorix; 52 pp. ISBN 80-85526-12-3.
- ↑ GREGOR, Pavel – WIDIMSKÝ, Petr, et al. Kardiologie. 2. edition. Prague : Galén, 1999. 595; 168 pp. ISBN 80-7262-021-5.
- ↑ KLENER, Pavel, et al. Vnitřní lékařství. 3. edition. Prague : Galén and Karolinum, 2006. 1158 pp. ISBN 80-7262-430-X.
- ↑ LIU, H. H. – GUO, J. B. – GENG, Y.. Procalcitonin: present and future. Irish Journal of Medical Science. 2015, y. 3, vol. 184, p. 597-605, ISSN 0021-1265. DOI: 10.1007/s11845-015-1327-0.
- ↑ ÚKBLD 1. LF a VFN Praha. Prokalcitonin: vývoj názorů na interpretaci [online]. ©2009. [cit. 2011-06-30]. <http://www.cskb.cz/res/file/akce/sjezdy/2009-Pha/ppt/B1/Kazda.pdf>.