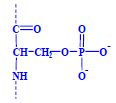
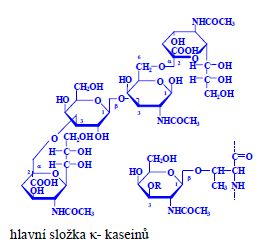
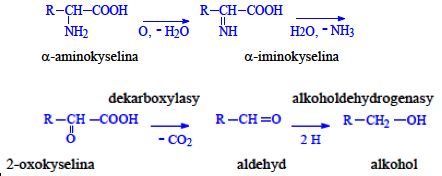
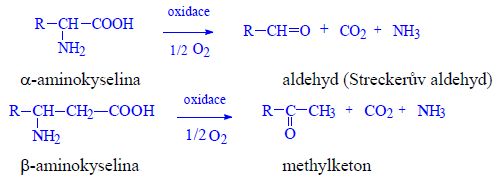
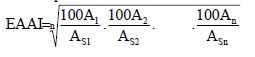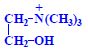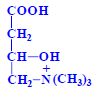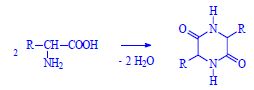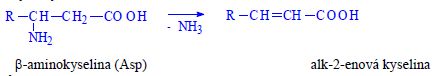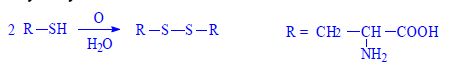Proteins (1. LF UK, NT)
Proteins, or polypeptides, are organic macromolecular substances. Their molecular weight exceeds 10,000. They consist of amino acids in numbers greater than 100. A typical protein contains 200-300.
Construction[edit | edit source]
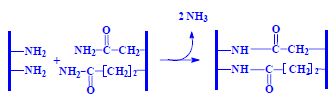
amino acids in the peptide are bound to each other by peptide binding. Peptide binding combines with simple covalent binding amino group of one amino acid and a carboxylic group of the other amino acid. The Gibbs energy value of this reaction is equal to G = 10 kJ/mol.
Polycondensation produces an arbitrarily long chain of amino acids. The end of the chain, which has a free (unreacted) amino group, is called the N-end. On the opposite side of the chain, on the other hand, we find a free carboxyl group. This end is called the C-end.
Structure[edit | edit source]
The structure of proteins is based on the arrangement of amino acids in the chain. Protein structure is very important for their function.
- Primary structure
The primary structure is defined by the exact sequence of amino acids in the chain.
- Secondary structure
Secondary structure means the spatial arrangement of amino acids in a chain and stabilization by hydrogen bonds.
- They are two basic secondary structures:
- α-helix: The string is curled into a right-hand helix. The length of a single screw thread is equal to 3,6 amino acid residues. The structure of α-helix is found primarily in fibrous proteins (keratins) or muscle proteins.
- β-folded sheet: Two parallel and antiparallel chains resembling a folded sheet of paper.
- Tertiary structure
The tertiary structure is characterized by other intramolecular binding interactions. For example, disulfide bridges, ionic bonds and van der Waals forces. Other H-bridges may also form in the molecule.
- Quarternary structure
The quaternary structure is formed in proteins consisting of two or more polypeptide chains. Their connections provide extramolecular binding interactions with each other. The quaternary structure is found, for example, in hemoglobin. In contrast, myoglobin does not possess quaternary structure.
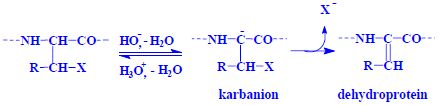
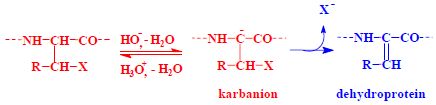
Protein denaturation[edit | edit source]
Protein denaturing is the process by which secondary and tertiary structures change. The protein thereby loses its biological activity. Denaturation can be achieved, for example, by heating or changing the pH.
Main nutrients[edit | edit source]
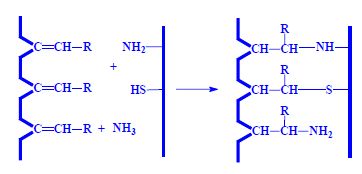
- peptide bonds
- other bindings
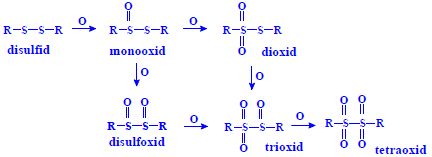
- disulphide -S-S-
- ester
- amide
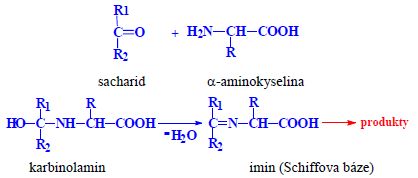
- Ingredients other than amino acids (physically or chemically)
- water
- inorganic ions lipids
- lipids, sugars, nucleic acid, colored compounds
| Clasification | |
|
| Meat, meat products, poultry, fish | |
|
| Egg | |
|
| Foods of vegetable origin | |
|
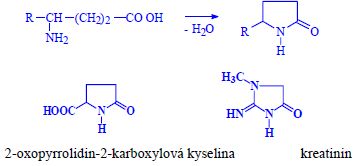
| Reactions | |
|
References[edit | edit source]
Related articles[edit | edit source]
Sources[edit | edit source]
- DAVÍDEK, Jiří. 2. AMINOKYSELINY, PEPTIDY, BÍLKOVINY [online]. [cit. 2022-12-30]. <https://connect.cuni.cz/p51525121/>.
- KOOLMAN, Jan – RÖHM, Klaus-Heinrich. Barevný atlas biochemie. 1. edition. Grada, 2012. ISBN 978-80-247-2977-0.
- MATOUŠ, Bohuslav, et al. Základy lékařské chemie a biochemie. 1. edition. Galén, 2010. 540 pp. ISBN 978-80-7262-702-8.
- MURRAY, Robert K. (Robert Kincaid) – BENDER, David A – BOTHAM, Kathleen M, et al. Harper’s illustrated biochemistry : [object Object]. 29. edition. McGraw-Hill Medical ; New York [u.a.] : McGraw-Hill Lange, 2012. ISBN 9780071765763.
- LEDVINA, Miroslav, et al. Biochemie pro studující medicíny. 2. edition. Karolinum, 2009. 548 pp. ISBN 978-80-246-1414-4.