Glucose Formation
Gluconeogenesis is the process of creating glucose molecules from substances of a non-carbohydrate nature. Precursors are mainly three- and four-carbon substances - e.g. lactate, pyruvate, glycerol, alanine, glycine and other glucogenic amino acids or propionate (mainly in ruminants). Gluconeogenesis is localized both in the matrix mitochondria and in the cytosol, mainly in liver cells, kidney tubular cells and enterocytes. Thanks to gluconeogenesis, we can survive even longer starvation, because glycogen reserves are exhausted within approximately 24 hours of starvation. However, it is activated already in the morning after a night's fasting.
The process of gluconeogenesis could take place simply by reversing the glycolysis. However, some reactions of glycolysis are irreversible and must be bypassed in gluconeogenesis with the use of different enzymes. We are talking about the so-called bypasses 1, 2 and 3. The three irreversible reactions in glycolysis are catalyzed by three kinases: pyruvate kinase, 6-phosphofructo-1-kinase and hexokinase/glucokinase.
Gluconeogenesis reaction[edit | edit source]
In the following overview, we will describe the individual reactions of gluconeogenesis:
- 1. Bypass 1 – conversion of pyruvate to phosphoenolpyruvate
- The first step is the transport of pyruvate into the mitochondrial matrix. This is followed by its carboxylation to oxaloacetate catalyzed by pyruvate carboxylase with simultaneous consumption of ATP (the cofactor is carboxybiotin). Oxaloacetate is then transported from the mitochondrion to the cytosol (transamination to Asp or reduction to malate), where the enzyme phosphoenolpyruvate carboxykinase converts it to phosphoenolpyruvate (GTP is consumed).
- 2. Reactions 3–8 are essentially the reverse form of glycolysis and take place in the cytosol
- 3. Bypass 2 – conversion of Fru-1,6-bisP to Fru-6-P
- Fructose-1,6-bisphosphatase hydrolyzes Fru-1,6-bisP to Fru-6-P. This reaction represents a key regulatory step in gluconeogenesis.
- 4. Fru-6-P isomerizes to Glc-6-P
- 5. Bypass 3 – conversion of Glc-6-P to free glucose
- Glucose-6-phosphatase hydrolyzes Glc-6-P to free glucose – thus catalyzing the cleavage of phosphate. This enzyme is bound to the membranes of the smooth endoplasmic reticulum. Glc-6-P is transported to the ER by the enzyme translocase. This separation into the ER serves to ensure that the resulting glucose is not immediately rephosphorylated to Glc-6-P. Free glucose is then released into the blood, where it can serve as a source of energy.
Energy balance of gluconeogenesis[edit | edit source]
Gluconeogenesis is an energy-intensive process – it consumes six macroergic phosphates per one molecule of glucose. In summary, we can express it by the following equation:
- 2 Pyr + 4 ATP + 2 GTP + 2 NADH + 4 H2O → Glc + 4 ADP + 2 GDP + 6 Pi + 2 NAD+ + 2 H+
Substrates for gluconeogenesis[edit | edit source]
- Lactate
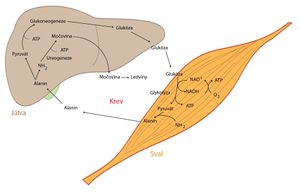
- Lactate, one of the main sources of carbon atoms in the process of gluconeogenesis, is produced during anaerobic glycolysis from pyruvate by a reaction catalyzed by lactate dehydrogenase (LDH). Its main producers are working muscle cells and erythrocytes. From them, lactate is released into the bloodstream, which takes it to the liver, where it is converted into glucose. Glucose is then released into the blood, from where the aforementioned cells can obtain it again. With this, we closed the so-called Cori cycle.
- Pyruvate
- Pyruvate can be produced by many peripheral tissues. At this point, we will describe the so-called glucose-alanine cycle, which takes place between muscle cells and the liver. After pyruvate is formed in muscle cells, it undergoes transamination to form alanine. It is released into the blood, which transports it to the liver, where alanine is converted back into pyruvate by transamination, which can be involved in gluconeogenesis. The resulting glucose is transferred by blood to the muscles and the whole cycle is closed.
- Glucogenic amino acids
- The carbon skeletons of all amino acids except leucineu and lysineu can be a source of carbon atoms for the process of gluconeogenesis. Alanine and glutamine are the main ones. The exact mechanism of their involvement is beyond the scope of this tutorial. The main source of glucogenic amino acids is muscle proteins.
- Glycerol
- Glycerol obtained during the hydrolysis of triacylglycerols can be used as a substrate for gluconeogenesis. The first step is its phosphorylation to glycerol-3-P by glycerol kinase. This is followed by its dehydrogenation to dihydroxyacetone-P catalyzed by glyceraldehyde-3-phosphate dehydrogenase, which gives rise to an intermediate of gluconeogenesis.
Energy[edit | edit source]
Energy for gluconeogenesis is mainly obtained from β-oxidation of fatty acids - during starvation, fatty acids are released from the stored triacylglycerols of adipose tissue and are metabolized in the liver.
Regulation of gluconeogenesis[edit | edit source]
Gluconeogenesis is a metabolic pathway that is activated mainly during starvation or in pathological conditions (stress due to infection, polytrauma, etc.).
Gluconeogenesis regulatory enzymes are those that bypass the irreversible reactions of glycolysis:
- Pyruvate carboxylase - it is activated by acetyl-CoA originating, for example, from the β-oxidation of fatty acids.
- PEP carboxykinase, Fru-1,6-bisphosphatase and Glc-6-phosphatase - they are regulated by the same influences as glycolysis reactions, only in the opposite direction. For example, Fru-1,6-bisphosphatase is activated by citrate, while AMP or Fru-2,6-bisP shows an inhibitory effect.
In addition to the activity of regulatory enzymes, an important factor determining the effectiveness of gluconeogenesis is the supply of its substrates, which are produced, for example, by proteolysis or lipolysis.
Counterregulatory hormones (glucocorticoids, glucagon or catecholamines) enhance gluconeogenesis, insulin, on the contrary, inhibits it.