Proteinuria
Physiologically, urine contains a very small amount of protein that is not detectable by basic precipitation or colorimetric techniques – we speak of so-called physiological proteinuria. If there is a larger amount of protein in the urine, it is proteinuria in the narrower sense of the word.
Physiological proteinuria[edit | edit source]
A healthy adult normally excretes about 50-80 mg of protein, maximum 96 mg/m2 (i.e. about 150 mg) in 24 hours (physiological proteinuria) in the urine during normal physical activity. About 40% of these proteins are plasma proteins and the remaining 60% are renal proteins (Tamm-Horsfall uromucoid) and urinary tract proteins (secretory IgA, unspecified glycoproteins, and glycopeptides).
The amount and composition of proteins in urine depend on their filtration through the glomerular membrane, resorption in tubular cells and renal haemodynamics.
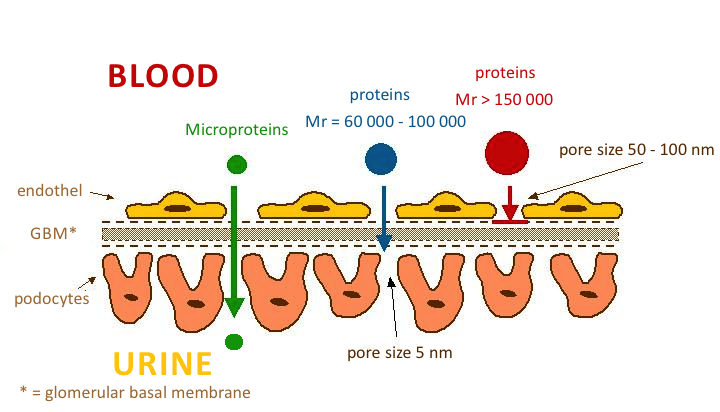
Normally, the glomerular membrane prevents the excretion of substances with a molecular weight of Mr > 60 000.
In glomerular ultrafiltration, two essential factors apply:
- The microporous structure of the glomerular basement membrane prevents the penetration of plasma proteins with Mr greater than 100 000-150 000 (e.g. IgG and IgA). We refer to size selectivity.
- Electrostatic barrier' is determined by the surface of all structural components of the glomerular wall. Hydrated polyanionic macromolecules of glomerular wall structures exhibit a predominant negative charge. Most plasma proteins also have a negative charge that causes them to be repelled. This mechanism prevents the permeation of proteins even with molecular weights around 60 000-70 000 (e.g. albumin and transferrin). Neutral or even positively charged molecules pass through the glomerular filter much more easily. This is charge selectivity.
Classification of proteinurias[edit | edit source]
The classification of proteinuria is of considerable diagnostic value. Proteinuria can be divided into three basic groups, which are further classified:
- Prerenal.
- Renal:
- glomerular proteinuria,
- selective,
- non-selective,
- tubularproteinuria,
- mixed-proteinuria.
- glomerular proteinuria,
- Postrenal.
Prerenal proteinuria[edit | edit source]
Prerenal proteinuria (overflow proteinuria) occurs at high plasma concentrations of low-molecular-weight proteins, which even under physiological circumstances can pass into the ultrafiltrate. Glomerular permeability of proteins may not be impaired. Tubular resorption may also be normal, but due to the increased load, some protein leaks into the urine because the resorption capacity of the proximal tubule is exceeded.
Prerenal proteinuria occurs e.g. in excessive intravascular haemolysis, when haemoglobin (haemoglobinuria) is present in the urine, or in crush syndrome and rhabdomyolysis, when myoglobin (myoglobinuria) is present in the urine. The presence of immunoglobulin light chains in urine (so-called Bence-Jones protein) is indicative of myeloma. Prerenal proteinuria often accompanies the onset of some acute inflammatory and necrotizing diseases. They are conditioned by the secretion of tissue degradation products and, to some extent, some low-molecular-weight acute phase proteins (e.g. orosomucoid).
Renal proteinuria[edit | edit source]
Renal proteinuria is divided according to the affected part of the nephron into glomerular, tubular, and mixed.
- Glomerular proteinuria
- is a manifestation of increased permeability of the glomerular wall to proteins. In selective glomerular proteinuria, when the negative charge of the glomerular membrane is lost, proteins of intermediate molecular mass with Mr between 70 000-100 000 (especially albumin and transferrin) are excreted into the urine at an increased rate, while proteins of high molecular mass are retained. In the urine, albumin predominates and has the highest negative charge. With more severe damage, the glomerular membrane loses the ability to discriminate proteins by size during filtration, and proteins with Mr above 100 000 such as IgG penetrate the urine in addition to proteins of intermediate size. We are talking about nonselective glomerular proteinuria. In glomerular proteinuria, usually the daily protein loss exceeds 2 g.
- Tubular proteinuria
- is caused by reduced tubular resorption of normally filtered proteins. It is characterized by increased secretion of low-molecular-weight proteins (microproteins) that are resorbed in the tubules under physiological conditions (β2-microglobulin, α1-microglobulin, free immunoglobulin light chains). A very common cause is the tubular damage by some nephrotoxic drugs (cytostatics, some antibiotics, many analgesics, and anti-inflammatory agents) or heavy metals (Hg, Pb, Cd). Tubular proteinuria may accompany some prerenal proteinuria (paraproteinuria, myoglobinuria) because there is a competitive relative lack of tubular resorption of other freely filterable proteins. Protein losses in tubular proteinuria are not large, usually 0.3-1.5 g/24 hours.
Mixed proteinuria is a combination of non-selective glomerular proteinuria and tubular proteinuria. It is a manifestation of the death of most nephrons.
Postrenal proteinuria[edit | edit source]
Postrenal proteinuria occurs in hemorrhage, tumors, and urinary tract infections when there is a direct passage of plasma or protein exudation into the urine. It is reliably identified by the detection of plasma macromolecular proteins of high molecular weight (α2-macroglobulin, IgM), which do not penetrate the wall of the glomerular capillary even in non-selective proteinuria.
| Type of proteinuria | Characteristic proteins in the urine | Notes |
|---|---|---|
| RENAL | ||
| Selective glomerular |
(Mr 70 000–100 000) |
|
| Non-selective glomerular |
(Mr > 70 000) |
|
| Tubular |
(Mr 10 000–70 000) |
|
| Mixed (tj. glomerulo-tubular) |
|
|
| PRERENAL |
|
|
| POSTRENAL |
|
|
Functional proteinuria[edit | edit source]
Functional (also benign) proteinuria is a special form of glomerular proteinuria. It is explained by changes in blood flow through the glomerulus. It is encountered after great physical activity, colds, in fevers, or congestive heart failure[1]. Protein losses do not exceed 1 g/day. In some adolescents, proteinuria occurs only in the upright position, so-called postural (orthostatic) proteinuria, which is often explained by vasoconstriction in lumbar hyperlordosis and is the most common cause of proteinuria in children[2]. In this case, daily protein losses can exceed even 1 g. Most of these proteinuria spontaneously adjust after the provoking cause has resolved. Also, in pregnancy, urinary protein waste increases to 200-300 mg/day.
Albuminuria[edit | edit source]
Urinary excretion of albumin does not physiologically exceed 30 mg/24 hours (i.e. 20 μg/min or about 15-20 mg/l)[3]. However, conventional tests for proteinuria (using diagnostic strips, sulphosalicylic acid test) can only detect protein when the albumin concentration exceeds about 150 mg/l, i.e. when it is practically 10 times elevated[4]. Losses of small amounts of albumin (30-300 mg/24 h) are demonstrable by immunochemical methods.
In older literature, the term microalbuminuria was used for small albumin losses that are demonstrable by immunochemistry but not by conventional proteinuria tests.
Screening for albuminuria is particularly valuable in patients with diabetes mellitus type II, but also in other disorders of glucose metabolism and in hypertensives. The finding of small amounts of albumin in the urine is an early sign of complications of these diseases, especially diabetic or hypertensive nephropathy and vasculopathy, and is often a reason to intensify treatment.
An increase in albuminuria is a very sensitive indicator of damage to the glomerular apparatus. This is due to the fact that albumin in small amounts passes through the glomerular membrane even physiologically. Under normal circumstances, however, it is almost completely resorbed in the proximal tubules. However, the tubular resorption capacity of albumin is virtually exhausted already during physiological albumin filtration; any increase in the concentration of this protein in the glomerular filtrate therefore leads to a rapid increase in the concentration of albumin in the final urine[4].
| Albuminuria | |||
|---|---|---|---|
| mg/24 hour | μg/min | mg/mmol of creatinine | |
| norm | < 30 | < 20 | < 3,5 |
| elevated albuminuria | 30–300 | 20–200 | 3,5–35 |
| detectable proteinuria | > 300 | > 200 | > 35 |
To monitor disease progression and manage treatment, albuminuria needs to be quantified more precisely. Albumin is determined in urine collected overnight and losses are converted to μg of albumin per minute. Values less than 100 μg/min usually correspond to reversible damage, which can be influenced by careful compensation for diabetes and arterial hypertension[3].
Another option is to determine albumin in the first morning urine sample and calculate the albumin/creatinine ratio. Physiologically, this index is around 2.8-22.8 g of albumin per mole of creatinine[4].
For albuminuria testing to be meaningful, it is necessary to exclude uroinfection.
Qualitative determination of protein in urine[edit | edit source]
Diagnostic strips are used to detect pathological proteinuria. In some laboratories, the strip test is combined with a sulfosalicylic acid test.
Diagnostic strips[edit | edit source]
The principle of protein determination in urine using diagnostic strips is based on the so-called protein acid-base indicator error, e.g. tetrabromophenol blue, tetrabromophenolphthalein ethyl ester, or 3´,3´´,5´´,5´´-tetrachlorophenol-3,4,5,6-tetrabromophenolphthalein. Like any acid-base indicator, these substances change colour at a certain pH (they behave like weak acids, with the protonated form having a different colour than the dissociated form): at pH lower than 3.5 they are yellow, at higher pH they are green to blue. In addition to the indicator, the reaction zone of the test strip contains a buffer which maintains the pH between 3,0 and 3,5, so the indicator is yellow. If there are proteins in the sample, they bind to the indicator with their amino groups. However, this changes its properties – the transition region shifts towards the more acidic pH. This means that at the constant pH between 3.0 and 3.5, the indicator with the protein bound will be green, as if it were in a more alkaline environment (hence the protein indicator error). The intensity of the colour depends on the concentration of the protein, ranging from green to blue, and is assessed visually or instrumentally.
For strongly alkaline urines (pH above 8) or if the urine is very concentrated, the test may give false positive results (buffer depletion in the reaction zone). In these cases, we acidify the urine with a few drops of dilute acetic acid to pH 5-6 and repeat the test. False positives can also be caused by high concentrations of certain substances with amino groups (contamination of the collection vessel by certain disinfectants), which bind to the indicators in a similar way to proteins.
The disadvantage of the test strips is their different sensitivity to individual proteins. The strips react very well with albumin and indicate its presence in urine from 0.1 to 0.5 g/l. They show significantly lower sensitivity to globulins, glycoproteins, and Bence-Jones protein. These diagnostic strips cannot demonstrate the increase in albuminuria to values up to about 200 mg/l or the daily albumin loss of 30 to 300 mg/24 hours that accompanies the earlier stages of some nephropathies. Immunochemical methods such as special diagnostic strips based on immunochromatographic principles or immunoturbidimetry can be used to screen for an increase in albuminuria.
Sulfosalicylic acid test[edit | edit source]
The principle of the test is denaturation of the protein by sulfosalicylic acid, which results in opalescence to opacity.
The reaction is sensitive, showing 0.1-0.2 g/l of total proteinuria. Differences in the detection of individual proteins are not as pronounced as with diagnostic strips. Sulfosalicylic acid also precipitates globulins.
False-positive results are given by this test when certain X-ray contrast agents, penicillin, sulphonamides, salicylic acid, and antidiabetic drugs are excluded.
A semi-quantitative scale is used for evaluation:
| Finding | Evaluation | Approximate concentration of protein in g/l |
|---|---|---|
| Opalescence | trace amounts | 0,05–0,1 |
| Light haze (transparent, underlying text is readable) | + | 0,1–0,2 |
| Milky haze (opaque, without flakes) | ++ | 0,5–1,0 |
| Milky haze with flake formation | +++ | 2,0–5,0 |
| Flaky clot | ++++ | ≥ 5,0 |
Quantitative determination of protein in urine[edit | edit source]
Quantitative determination of protein in urine is methodologically quite difficult. In clinical-biochemical practice, methods are used for the determination of proteinuria, which can be divided into three groups according to the principle:
- methods based on denaturation followed by turbidimetric determination of the haze (e.g. methods with trichloroacetic acid or sulfosalicylic acid);
- colorimetric methods with or without prior denaturation of proteins (e.g. biuret reaction);
- methods based on binding of dyes to proteins (e.g., the method with Coomassie brilliant blue G 250 according to Bradford, with pyrogallol red, etc.).
Today, techniques that can be automated - turbidimetry and colour reaction with pyrogallol red - are preferred.
Determination of proteinuria using pyrogallol red[edit | edit source]
Pyrogallol red is used for protein quantification, e.g. for the determination of proteinuria. Pyrogallol red forms a pink complex with molybdenum with an absorption maximum at 470 nm. When the protein binds to this complex in an acidic environment, the colour deepens and the absorption maximum shifts to the 600 nm region. The absorbance of the resulting complex of reagent with protein is linearly dependent on the concentration of protein in the sample in the concentration range 0.06-2.0 g/l.
Assessment of the magnitude of proteinuria[edit | edit source]
| < 0.150 g/24 hod | physiological proteinuria |
| < 1 g/24 hod | low proteinuria (mostly tubular) |
| 1.0–3.5 g/24 hod | medium proteinuria |
| > 3.5 g/24 hod | high proteinuria |
| > 10 g/24 hod | proteinuria usually associated with severe nephrotic syndrome |
Typification of proteinuria[edit | edit source]
To determine the type of proteinuria, it is necessary to know the spectrum of proteins excreted in the urine. Electrophoretic methods are used for this purpose. Electrophoretic partitioning of urinary proteins according to their molecular weight allows semiquantitative evaluation of individual diagnostically significant proteins and classification of proteinuria. Electrophoresis in agarose or polyacrylamide gel has gradually become the method of choice for urinary protein analysis.
In order to separate the proteins by size (rather than charge), a polyacrylamide gel can be used, the density of which increases from the cathode to the anode (i.e., the mesh or pore size in the gel gradually decreases). Small molecules in such a gel travel further than large molecules.
Another, more commonly used option, is to treat the sample with the detergent sodium lauryl sulfate (sodium dodecyl sulfate – SDS), which "surrounds" the protein and replaces its own charge with its negative charge. The resulting complexes have approximately the same charge (more precisely: they have the same surface charge density). If electrophoresis is then carried out in a relatively dense gel, it is separated according to relative molecular weight: smaller molecules travel through the gel faster than large ones (molecular sieve technique). It moves the fastest β2-microglobulin, albumin lies about midway along the partition pathway; between the start and albumin are proteins with Mr greater than 70 000.
Urinary protein electrophoresis evaluation[edit | edit source]
In glomerular proteinuria we find proteins in the electrophoreogram between start and albumin (i.e. Mr > 70 000).
Proteins observed in glomerular proteinuria Mr Albumin 68 000 selective non-selective Transferrin 77 000 selective non-selective IgG 150 000 non-selective IgA 160 000 non-selective Haptoglobins 85 000–1 000 000 non-selective
Tubular proteinuria are characterized by the presence of protein between albumin and the anodic end of the electrophoreogram (i.e. Mr < 70 000).
Proteins observed in tubular proteinuria Mr β2-microglobulin 11 800 Lysozyme 15 000 Retinol binding protein (RBP) 21 000 Ig free light chains 25 000 α1-microglobulin 33 000 Dimeric Ig free light chains 50 000 Albumin 68 000
Proteins observed in tubular proteinuria
Presence of α2-macroglobulin (Mr = 800 000) with other findings is similar to mixed proteinuria suggest postrenal proteinuria.
Links[edit | edit source]
Related articles[edit | edit source]
External links[edit | edit source]
- ↑ BURTIS, Carl A – ASHWOOD, Edward R – BRUNS, David E. Tietz textbook of clinical chemistry and molecular diagnostics. 4. edition. St. Louis, Mo : Elsevier Saunders, 2006. 2412 pp. pp. 576. ISBN 978-0-7216-0189-2.
- ↑ MUNTAU, Ania Carolina. Pediatrics. 4. edition. Grada, 2009. pp. 420. ISBN 978- 80-247-2525-3.
- ↑ a b ZIMA, Tomáš, et al. Laboratorní diagnostika. 2. edition. Praha : Galén a Karolinum, 2007. 906 pp. pp. 106-7, 121-2. ISBN 978-80-246-1423-6.
- ↑ a b c RACEK, Jaroslav, et al. Klinická biochemie. 2. edition. Praha : Galén, 2006. 329 pp. pp. 170. ISBN 80-7262-324-9.
- ↑ KRAML, Jiří, et al. Návody k praktickým cvičením z lékařské chemie a biochemie. 1. edition. Praha : Karolinum, 1991. 312 pp. pp. 243. ISBN 80-7066-453-3.