Direct diagnosis of hereditary diseases by nucleic acid analysis
It makes it possible to capture and identify the mutation responsible for the disease in the affected and in the monitored family.
The condition is knowledge of the location of the gene and knowledge of its standard sequence. They offer the possibility of detecting a heterozygous carrier of a mutated allele even in an individual in whose family the affected individual is unknown. The investigation begins in the sections of the gene with the highest number of mutations.
Methods that pick up any deviation from the standard DNA sequence[edit | edit source]
Most methods use PCR for detection. Most genes cannot be examined in one reaction due to their size. Using selected primers, individual sections of the gene are gradually amplified.
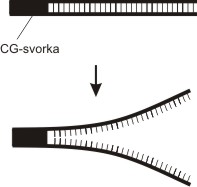
Heteroduplex Analysis[edit | edit source]
Based on detection of incorrect pairing of bases (mismatch). This occurs during the hybridization of the complementary DNA strand of the standard and mutant type, when hybrid DNA molecules - heteroduplexes - are created. They are formed at sites with deletions or insertions of several bases. The principle is the different electrophoretic mobility'' of dsDNA in the case of perfect and imperfect complementarity.
DGGE[edit | edit source]
Denaturing Gradient Gel Electrophoresis. It is based on a different melting point' depending on the composition of the dsDNA. Heteroduplexes and homoduplexes – they migrate during gel electrophoresis with a linearly increasing amount of denaturing agents. At the point where the concentration of the denaturing agents corresponds to the temperature at which partial denaturation of DNA occurs, the migration of the partially denatured section of DNA is greatly slowed down or stopped. DNA heteroduplexes are less stable and therefore their strands partially separate earlier than the strands of homoduplexes - they slow down their mobility in the gel and migrate to a different position.
High Resolution Melt[edit | edit source]
Detection of mutations and polymorphisms in dsDNA. The advantage of the method is speed and cheapness.
Procedure: PCR - HRM analysis = increasing the temperature to the melting temperature, which leads to the separation of the DNA strands. The process can be captured in real time. Binding of fluorescent dye to DNA.
Chemical or enzymatic cleavage of heteroduplexes[edit | edit source]
Mismatch cleavage technique. Single-stranded site cleavage - where there is not complete complementarity of both strands. Electrophoresis in a denatured gel - proof of the presence of cleaved or uncleaved fragments of the tested DNA.
Capturing large deletions[edit | edit source]
Deletions affecting an entire exon or gene are not identifiable by conventional methods. They can be detected using Southern blotting methods. In the case of extensive deletions, the FISH method can be used.
Real time PCR[edit | edit source]
Determining the exact number of copies of certain nucleic acid sequences in cells. It allows to monitor the amount of DNA product in each PCR cycle. E.g. when detecting the level of viral infection, the number of oncogene copies.
DNA Chips[edit | edit source]
The most promising of modern methods. Can be used in combination with computerized evaluation of results.
The following methods are no longer used!
SSCP method[edit | edit source]
'Single Strand Conformation Polymorphism Analysis. Based on ssDNA (single-stranded DNA) analysis, it uses PCR. The amplified DNA segment is denatured and applied to a polyacrylic gel. If a mutation is present in the investigated section of DNA, the single-stranded DNA takes a different conformation (different mobility of this DNA in the gel). The fibers can be visualized - radioactive labeling of oligonucleotides for PCR.
RT-PCR[edit | edit source]
The starting material is isolated RNA, which is converted into cDNA using reverse transcriptase and amplified by PCR. It is then tested for the presence of mutations. It allows you to test larger sections. Capture of aberrant splicing' or activation of cryptic site of splicing that are very difficult to identify by conventional methods.
PTT[edit | edit source]
Protein Truncation Test. A method specific for the detection of mutations that result in the formation of a "premature termination codon" and thus a shortening of the protein product. cDNA is prepared from the isolated mRNA by reverse transcription and subsequent PCR, then transcription and translation in vitro are performed. The size of the protein product was tested electrophoretically and compared to the length of the standard product. Used for frameshift and nonsense mutation detection.
DNA Sequencing[edit | edit source]
Use especially in the final phase of the examination. It detects deviations in the nucleotide sequence of DNA.
Sanger method[edit | edit source]
The most frequently used.
The basis is the use of a mixture of standard deoxy nucleotide triphosphates (dNTPs) and modified dideoxy nucleotide triphosphates (ddNTPs) in the preparation of the DNA segment to be sequenced. Modified ddNTPs result in termination of DNA synthesis. The result of ``DNA synthesis is a series of DNA molecules of different lengths, each differing in length by ``one nucleotide.
The whole process takes place in 4 separate enzyme reactions. Each contains - template DNA in single-stranded form, DNA-polymerase, primer, 4 dNTPs and a modified nucleotide dideoxy ``nucleotide triphosphate (ddNTPs). In each of the 4 reactions there is a different ddNTP - ddATP, ddGTP, ddCTP, or ddTTP.
After sequencing, the DNA molecules are separated by capillary electrophoresis - the products pass through the capillary, where they are separated according to size. If radioactively labeled dNTP is added to the sequencing reaction - it is possible to detect the resulting DNA on x-ray film in the form of visible bands of all 4 lines.
Methods of detection of specific mutations[edit | edit source]
They can be used where the majority of affected individuals in the population have one or a limited number of mutations' E.g. sickle cell disease, cystic fibrosis.
Mutation-specific RFLP analysis[edit | edit source]
In the case where the mutation creates or destroys a restriction site' for a certain restriction enzyme and is the cause of the disease at the same time. The PCR method is used to amplify a section of DNA with a restriction site. This is followed by restriction with specific restriction enzymes and gel electrophoresis.
ASO[edit | edit source]
Alele Sspecific O oligonucleotides. Synthesized to detect a point nucleotide difference in a DNA sequence.
Trinucleotide expansion diagnosis[edit | edit source]
DNA diagnostics in the case of Huntington's disease. Amplification (PCR) in the region of the gene with a high content of repeats (CAG)n. This is followed by electrophoresis in a polyacrylamide gel. 'The number of repetitions is related to the development of the disease. In the detection of Fragile X Syndrome, it is possible to use PCR to detect standard or premutant alleles. Full mutations are too long.
Links[edit | edit source]
Related Articles[edit | edit source]
Source[edit | edit source]
- {{#switch: web
|book =
Incomplete publication citation. ŠTEFÁNEK, GeorgeAlso available from <https://www.stefajir.cz/>.
|collection =
Incomplete citation of contribution in proceedings. ŠTEFÁNEK, George. Also available from <https://www.stefajir.cz/>. {{ #if: |978-80-7262-438-6} } |article = Incomplete article citation. ŠTEFÁNEK, George. Medicine, diseases, studies at the 1st Faculty of Medicine, UK. also available from <https://www.stefajir.cz/>.
|web =
ŠTEFÁNEK, George. Medicine, diseases, studies at the 1st Faculty of Medicine, UK [online]. [cit. 2009]. <https://www.stefajir.cz/>.
|cd =
ŠTEFÁNEK, George. Medicine, diseases, studies at the 1st Faculty of Medicine, UK [CD/DVD]. [cit. 2009].
|db =
Incomplete database citation. Medicine, diseases, studies at the 1st Faculty of Medicine, UK [database]. [cit. 2009]. <https://www.stefajir.cz/>.
|corporate_literature =
Incomplete citation of company literature. ŠTEFÁNEK, George. Also available from <https://www.stefajir.cz/>. legislative_document = Incomplete citation of legislative document. Also available from URL <https://www.stefajir.cz/>.
References[edit | edit source]
- {{#switch: book
|book =
Incomplete publication citation. KOHOUTOVÁ, Milada. Medical biology and genetics (Part II). Prague : Karolinum Publishing House, 2013. 202 s. 978-80-7262-438-6.
|collection =
Incomplete citation of contribution in proceedings. KOHOUTOVÁ, Milada. Medical biology and genetics (Part II). Prague : Karolinum Publishing House, 2013. 202 s. {{
#if: 978-80-246-1873-9 |978-80-7262-438-6} }
|article =
Incomplete article citation. KOHOUTOVÁ, Milada. 2013, year 2013,
|web =
Incomplete site citation. KOHOUTOVÁ, Milada. Karolinum Publishing House, ©2013.
|cd =
Incomplete carrier citation. KOHOUTOVÁ, Milada. Karolinum Publishing House, ©2013.
|db =
Incomplete database citation. Karolinum Publishing House, ©2013.
|corporate_literature =
KOHOUTOVÁ, Milada. Medical biology and genetics (Part II). Prague : Karolinum Publishing House, 2013. 202 s. 978-80-7262-438-6} }