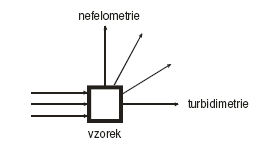
Turbidimetry
In addition to absorption, when light passes through a sample, it can be scattered when it comes to dispersive or colloidal system. This is used in the techniques of nephelometry (Greek nefele = cloud), when the intensity of light scattered at a certain angle is measured, and turbidimetry (English turbid = cloudy), when the intensity of light is measured passing through the sample in the original direction. Both methods are "mirror" of each other, therefore we will only deal with turbidimetry in the following: it can be carried out using the usual photometric equipment and the loss of light by scattering when passing through the sample can be easily described using absorbance and other quantities usual in photometry.
The amount of diffused light depends on
- particle concentration. In a wide range of concentrations, the dependence is linear, so it is possible to work completely analogously to photometric methods using the Lambert-Beer law. The quantity corresponding to absorbance is called turbidity.
- of particle sizes. The amount of light scattered is approximately inversely proportional to the molecular weight of the particle.
- wavelength of light. The shorter the wavelength, the greater the proportion of light will be scattered (Tyndall effect), the intensity of the scattered light increases approximately with the fourth power of the reciprocal of the wavelength. In practice, wavelengths from 340 to 450 nm are used as the most advantageous. Shorter-wavelength light is usually absorbed by proteins, which are usually present in biological samples.
With turbidimetric methods, it is usually the most difficult to create a sufficiently stable suspension that would be constant throughout the measurement period. That is why protective colloids, most often polyethylene glycol, are added to the reaction mixtures.
Turbidimetry and nephelometry are used most often in immunochemical methods for evaluation immunoprecipitation reactions, where the turbidity is formed by antigen-antibody complexes.