Topology of DNA
In vivo dsDNA tends to be a predominantly closed structure; viral, bacterial and mitochondrial genomes are often circular molecules, and eukaryotic chromosomal DNA is folded into loops, the ends of which are fixed on a protein complex. These closed dsDNA structures can be coiled in a second-order superhelix. If we consider DNA conformation at this level of structure, we speak of DNA topology.
Helix winding[edit | edit source]
If one end of a linear right-handed double helix is fixed and the other is rotated so that the winding of the helix "tightens", i.e. counter-clockwise, the DNA twists into a positive (left-handed) superhelix (of a superhelix). By rotating the fixed dsDNA in a clockwise direction, it can coil into the negative superhelix that is normally found in cells. The energy stored in the negative superhelix facilitates the unfolding of the dsDNA strands during replication. In it, the parental dsDNA rotates counterclockwise and the negative superhelix unwinds. This phenomenon can be well demonstrated on the rotation and coiling of a longer and thicker rubber.
Numerical description of the winding[edit | edit source]
Changes in the topological (and energetic!) states of DNA are expressed numerically:
- Total winding number L (linking number) indicates how many times one DNA strand is wound around another in the structure under consideration, regardless of whether in a helix or a superhelix.
- The twisting number T is the number of turns of the chains in the helix.
- The writhing number W indicates how many times the double helix crosses as a whole.
It follows that L = T + W. DNA molecules that differ only in the L number are topoisomers. In practice, changes in crossing are assessed rather than their absolute number, i.e. AL, AT, AW.
Topoisomerases[edit | edit source]
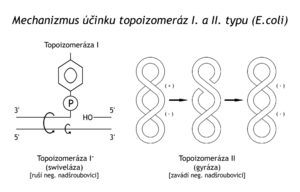
Topoisomerases are enzymes that introduce or break a superhelix.
Topoisomerase I[edit | edit source]
- Topoisomerases I. type creates a break on one of the chains, whereby the 5'-end of the break binds to the –OH group of the tyrosine residue of the enzyme. Any tension in the superhelix is released by rotating the helix around the s–bonds of the intact chain. Then the enzyme heals the broken chain again without supplying energy. The energy for strand rebinding is conserved in binding the 5'-end of the break to the enzyme. Topoisomerases I have been described in both bacteria and animal cells. An example is the ω-protein or swivelase in E. coli. Topoisomerase I breaks the negative superhelix.
Topoisomerase II[edit | edit source]
- Type II topoisomerases cleave both dsDNA strands at the same time, move another, intact section of dsDNA to the other side of the helix at the point of interruption, and heal both strands again.
An example of these enzymes is gyrase in the bacterium E. coli. A certain degree of supercoiling of DNA, required for the unfolding of the double helix, is thus ensured in the cell by opposing enzymes: topoisomerase I unfolds the negative superhelix, while gyrase, on the contrary, introduces it.
The negative superhelix has a higher free energy than relaxed dsDNA. Gyrase obtains this energy by splitting ATP.
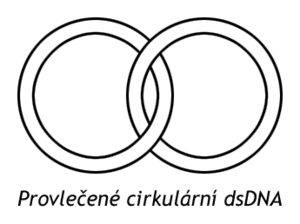
The binding of DNA strands to the enzyme is inhibited by nalidixic acid (an antibiotic), and the binding of ATP is inhibited by the antibiotic novobiocin. By introducing a negative superhelix of topoisomerase II. type help unfold the DNA helix at the replication fork. They also catalyze the release of interlaced circular dsDNA formed after replication of the viral genome.
Links[edit | edit source]
Related articles[edit | edit source]
- Structure of Nucleic Acids
- Basic components of nucleic acids
- Primary structure of nucleic acids
- Nucleic acid cleavage by hydrolysis
- Sequencing Methods
- Secondary structure of DNA
- Nucleic acid denaturation, molecular hybridization
- RNA Secondary Structure
- Interaction of DNA with proteins
- Bacterial Chromosome
- Eukaryotic chromosomes
- Mitochondrial DNA
Template:Brief Biochemistry (Pinch)
Resources[edit | edit source]
- ŠTÍPEK, Stanislav. Brief biochemistry : Storage and expression of genetic information. 1. edition. Medprint, 1998. 92 pp. pp. 19–20. ISBN 80-902036-2-0.