Tests for the Acid-base Balance Status
From WikiLectures
Clinical assesment[edit | edit source]
- The most clinical useful information comes from the clinical description of the patient by the history and physical examination.
- The H&P usually gives an idea of what acid base disorder might be present even before collecting the ABG sample.
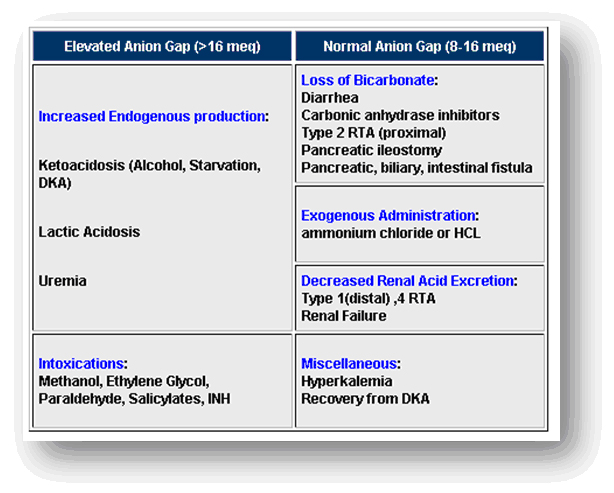
Agap:
- The major causes of simple and mixed disturbances shoud be kept in mind so that such disorders may be predicted from the patient’s clinical picture. For example,
- patients with severe vomiting and those on long-term thiazide therapy can be expected to have metabolic alkalosis,
- those with renal failure may have metabolic acidosis
- and patients with pneumonia or congestive cardiac failure may have respiratory alkalosis…
- The possibility of an acid-base balance disturbance suggested by clinical presentation should then be evaluated by appropriate laboratory tests.
Blood gases[edit | edit source]
- In order to interpret acid-base disturbances, the following five factors are considered:
- pH
- HCO3-
- PCO2
- Anion gap
- and assessment for compensation
- The first step is to determine if the patient is acidemic or alkalemic, based on pH.
- Second, the primary disorder is determined by evaluating HCO3– and PCO2.
- If HCO3– is elevated and pH is elevated, there is metabolic alkalosis.
- If both are decreased, there is metabolic acidosis.
- Next, one must look at the PCO2 in the context of the HCO3–.
- If HCO3– is within the normal reference range and PCO2 is elevated but the patient is acidotic,
- the condition is respiratory acidosis.
- If bicarbonate is within the normal reference range and PCO2 is decreased but the patient is alkalotic,
- the condition is respiratory alkalosis.
- If HCO3– is within the normal reference range and PCO2 is elevated but the patient is acidotic,
- Next determine the anion gap, using standard formulas below, to determine the etiology of metabolic acidosis.
- Anion gap = [Na] – ([Cl-] + [HCO3-])
- Finally the pH, HCO3–, and PCO2 are considered to determine if compensation is as expected based on the typical ratio of 20:1 for bicarbonate to carbonic acid.
- For example, both decreased HCO3– and PCO2 should produce a slightly decreased or nearly normal pH if they are in metabolic acidosis compensation.
- To determine the actual ratio of bicarbonate to carbonic acid, PCO2 is converted to H2CO3 using the relationship
- PCO2 * 0.03 = H2CO3
- Compensation for metabolic acidosis or alkalosis is achieved initially by the respiratory system.
- Respiratory compensation for acidosis means that the lungs increase the level of alveolar ventilation, which raises the pH toward normal.
- The increased ventilation eliminates or blos off CO2, which eliminates carbonic acid.
- Also, the presence of acidosis normally increases respiratory drive.
- The respiratory system compensates for a metabolic defect.
- Respiratory compensation for acidosis means that the lungs increase the level of alveolar ventilation, which raises the pH toward normal.
- In metabolic alkalosis, some decrease in ventilation occurs but the PCO2 generally remains normal since respiratory compensation doesn’t occur until alkalosis has been severe and prolonged.
- Compensation for metabolic alkalosis is less complete since hypoventilation is not a naturally sustainable condition.
- In metabolic alkalosis, some decrease in ventilation occurs but the PCO2 generally remains normal since respiratory compensation doesn’t occur until alkalosis has been severe and prolonged.
References[edit | edit source]
Acid-Base Disorders by Walmsley Koay and Watkinson
Clinical chemistry (A laboratory perspective) by Wendy Arneson and Jean Brickell