Quantum numbers
Quantum numbers describe the behavior of a quantum system. Mathematically, these are the parameters of the solution of the Schrödinger equation of the respective system. This solution is usually denoted by Ψ(r,t) and called the wave function. The square of the wave function has the meaning of the electron probability density.[1]
Atomic orbital[edit | edit source]
The wave function Ψ describing a particular electron of a particular atom is referred to as an atomic orbital (AO); the same expression is also used in the figurative sense of the space in which the electron is most likely to be found. The electron goes around the nucleusit moves along a trajectory which, however, due to Heisenberg's uncertainty principle, cannot be sufficiently determined. By choosing a certain level of probability, the part of space in which the electron will be located with the given probability can be defined and mathematically described using the wave function. For the sake of simplicity, this description is not presented in the form of specific wave functions, but only in the form of parameters, after substituting into the general solution, we would obtain a specific wave function. The parameters are called quantum numbers.
Individual quantum numbers[edit | edit source]
The solution of the Schrödinger equation (and thus the wave function Ψ and the atomic orbital) is completely determined by the three quantum numbers n, l, m. In addition, an electron in a given orbital can acquire two quantum states, which is why it is described by yet another quantum number m s. The electron itself is assigned a quantum number s among elementary particles.
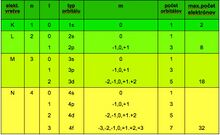
Principal quantum number n[edit | edit source]
The main quantum number (n =1,2,3,...) determines the total energy of the electron and thus also the shell in which the electron is located (K for n = 1, L for n = 2, M for n = 3, N for n = 4,...). For the energy of the electron, the relation applies:
Since the mass of the electron m, the charge of the electron e, the permittivity of the vacuum ε 0 and Planck's constant h, they can be summed up in a single constant k and the relationship can be written as:
Secondary quantum number l[edit | edit source]
The secondary quantum number (l =0,…(n −1)) is related to the quantization of the orbital angular momentum L. It determines the shape and symmetry of the electron cloud, i.e. the subshells s, p, d, f, g and h.
The constant ħ is called the reduced Planck constant and applies to it:
Magnetic quantum number m l[edit | edit source]
The magnetic quantum number (m l =0, ±1, ±2, … ± l) determines the orientation of the orbital in space; the magnetic orbital moment (the motion of an electric charge creates a magnetic field) has the opposite direction to the orbital angular momentum.
Spin quantum number with[edit | edit source]
The spin quantum number of electrons as well as protons and neutrons always has the value ½, which classifies these particles as fermions. In the case of an electron, the angular momentum given by its spin number is given by:
The spin of a charged fermion also manifests itself as its magnetic moment. It was the idea that a rotating electron actually represents a moving charge around which a magnetic field is created that led to this number being named spin. One of the fundamental properties of the microworld, which deviates from common macroscopic analogies, is quantization. If, for example, an electron is inserted into an external magnetic field, its magnetic moment cannot take any orientation. Only two orientations are possible, by taking them into account two values of the spin magnetic number are possible: +½ and −½., i.e. the projection into the external magnetic field can have only two values:
Pauli exclusion principle[edit | edit source]
The Pauli exclusion principle (also Pauli exclusivity principle) states that two fermions, i.e. fermions with identical quantum numbers, cannot exist in one quantum state at the same time. The principle is named after the Swiss physicist Wolfgang Pauli (1900–1958).
Since electrons are fermions, the Pauli exclusion principle also applies to them. The electron-specific formulation states that there cannot be two electrons in one atom that have all the same quantum numbers, i.e. they must differ by at least one quantum number. Among other things, this gives the maximum possible number of electrons in each shell.
Allowed transitions[edit | edit source]
During the transition of an electron from one energy level to another, absolutely arbitrary transitions are not possible. Only those transitions are possible (so-called allowed) when the main quantum number n changes arbitrarily, but the secondary quantum number l only changes by one. Other transitions are designated as forbidden, the probability of their occurrence is significantly lower than the probability of an allowed transition.
Footnotes[edit | edit source]
- In fact, it is a square only in the special cases where the Schrödinger equation has a real solution. If the solution is a complex function, it is the product of Ψ and functions complexly associated with it.
- Fermions are elementary particles with half-integer spin (ie their spin is an odd multiple of one-half). Their main characteristic is that they obey the Pauli exclusion principle. They are named after the Italian physicist Enrico Fermi (1901–1954). Their "opposite" are bosons, named after the Indian mathematician and physicist Satyendra Nath Bose (1894–1974), which do not obey the Pauli exclusion principle. An integer spin is usual for bosons. Typical bosons are e.g. photons.
- The reason for the non-zero probability of a forbidden transition is the existence of the tunnel effect. In this case, the latter allows the electron to make a "double jump" through the allowed intermediate state with a small but non-zero probability, even if it does not have enough energy for it.
Links[edit | edit source]
Literature[edit | edit source]
- HRAZDIRA, Vojtěch – MORNSTEIN,. Lékařská biofyzika a přístrojová technika. 1. edition. Brno : Neptun, 2001. 396 pp. ISBN 80-902896-1-4.
- MUCK, Alexander. Základy strukturní anorganické chemie. 1. edition. Praha : Academia, 2006. 508 pp. ISBN 80-200-1326-1.
Related articles[edit | edit source]
External links[edit | edit source]
- KUBATOVA, Senta. Biofot [online]. [cit. 2011-01-31]. <https://uloz.to/!CM6zAi6z/biofot-doc>.
- JANEČEK, Ivan – KALUS, René – HRIVŇÁK, Daniel. Kvantová, atomová a jaderná fyzika [online]. Katedra fyziky PřF OSU, [cit. 2011-09-21]. <http://artemis.osu.cz/mmfyz/index.htm>.
References[edit | edit source]
- ↑ MUCK, Alexander. Základy strukturní anorganické chemie. 1. edition. Praha : Academia, 2006. 508 pp. pp. 36. ISBN 80-200-1326-1.