Metabolism of the carbon skeleton of amino acids of the 2-oxoglutarate group, amino acids, branched-chain amino acids
- 2-oxoglutarate: arginine, histidine, proline, glutamine, glutamic acid
- succinyl-CoA: methionine, valine, threonine, isoleucine
- branched chain: valine, leucine, izoleucine
2-oxoglutarate[edit | edit source]
Arginine[edit | edit source]
- semi-essential: must be ingested with food during the development of children
- synthesized in the urea cycle and in the kidneys due to insufficient arginase activity
citrulline + aspartate + ATP --> argininosuccinate + AMP + PPi
argininosuccinate --> arginine + fumarate
- participates in the synthesis of creatine, nitric oxide
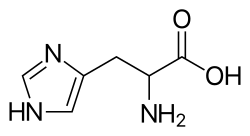
Histidin[edit | edit source]
- essential
- metabolized by non-oxidative deamination by histidase to uroconate --> glutamate --> 2-oxoglutarate
- substrate for the formation of carnosine (along with beta -alanine) and anserines - dipeptides in muscles that activate myosin ATPase
- decarboxylation produces histamine - in mast cells and in basophil granules
Proline[edit | edit source]
- non-essential amino (imino) acid
- is formed from 2-oxoglutarate in the presence of ATP and NADH to form 5-glutamate semialdehyde, which spontaneously cyclizes to pyrroline-5-carboxylate -> hydrogenation produces proline
- after transamination of glutamate semialdehyde --> ornithine
- in connective proteins; hydroxylation of proline increases rigidity (mainly in collagen fibers)
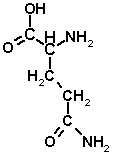
Glutamine[edit | edit source]
- non-essential
- to glutamate (--> 2-oxoglutarate) and ammonia by the action of glutaminase
Glutamic acid (glutamate)[edit | edit source]
- non-essential, key for AMK metabolism
- participates in transamination, glutamate dehydrogenase, glutamine synthetase and glutaminase reactions
- synthesis and degradation by transamination
- glutamate is a source for the synthesis of glutathione and GABA
Succinyl-CoA[edit | edit source]
Methionine[edit | edit source]
- essential
- in excess, the carbon skeleton is used as a source of E or for gluconeogenesis, sulfur is preserved in cysteine
- reaction catalyzed by methionine adenosyltransferase, all phosphate groups are released and S-adenosylmethionine or active methionine capable of methylating other substances is formed
- after the release of methyl groups from SAM, S-adenosylhomocysteine is formed, which is divided into homocysteine
- after combining homocysteine with serine --> cystathione, which is split into alpha- ketoglutarate, cysteine and ammonia
- resynthesis of methionine by homocysteine methyltransferase with cofactor vit. B12 (cobalamin) and methyl groups come from N-methyltetrahydrofolate
Valine, Leucine, Isoleucine[edit | edit source]
- essential, branched
The first 2 reactions are identical - first, transamination produces an alpha -keto acid, which undergoes oxidative decarboxylation by the mitochondrial complex to form acyl-CoA --> dehydrogenation to form unsaturated thiolacyl CoA --> NADH and CO2 are produced
- aminotransferases in 3 isoenzymes in the cytosol and mitochondria, 2 of which are capable of transaminating all 3 AMA, 1 is specific for leucine
Furthermore, a reaction that is similar to valine and isoleucine --> the formation of propionyl-CoA and acetylCoA (propionyl is converted into succinyl Coa).
Leucine is converted through a cascade of reactions to acetate and acetyle-CoA.
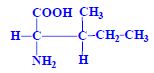
Threonine[edit | edit source]
- essential, polar with 2 chiral carbons
- breaks down by threoninaldolase into glycine and acetaldehyde --> acetate and acetyle-CoA
Links[edit | edit source]
Related articles[edit | edit source]