Gibbs energy (FBLT)
The difference in chemical potentials of products and reactants is related to a thermodynamic function called change in Gibbs energy (ΔG). Of the many possible definitions of Gibbs energy, two are important for biochemistry:
- The change in Gibbs energy is equal to the maximum amount of (non-volumetric) work the system can do;
- The change in Gibbs energy is a measure of the deviation from the equilibrium state.
The first definition tells us that we can use ΔG to predict whether a reaction will occur and whether it can be used as a source of energy for other processes (muscle contraction, movement of ions across a membrane).
The second definition expresses two facts:
a) in equilibrium state, ΔG is equal to zero;
b) changing the concentrations of substances in a system that is not in equilibrium will change (increase or decrease) ΔG.
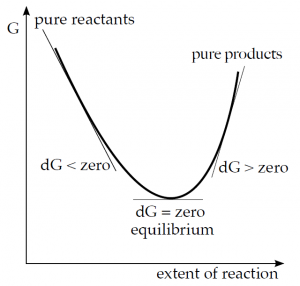
The meaning of the Gibbs energy change can also be illustrated by the following representation:
If a reaction starts with only a mixture of reactants, it will proceed towards products as long as 'ΔG is negative. If the chemical potential of the products becomes the same as that of the reactants, the system has reached the lowest energy state and the reaction stops. The reaction mixture is now at equilibrium. It cannot be assumed that the reaction would continue because ΔG is positive in the next part of the curve.
It therefore applies:
ΔG < 0 – the reaction takes place spontaneously; ΔG = 0 – 'equilibrium state; ΔG > 0 – the reaction does not take place spontaneously.
We can also define the change in Gibbs energy through thermodynamic quantities such as enthalpy (H) and entropy (S):
| (5) |
- ΔG = ΔH – T · ΔS
Enthalpy change is the energy released or consumed during a reaction (a negative value means energy is released and vice versa).
Entropy' is often considered to be a measure of the disorder of a system. According to the second law of thermodynamics, disorder (and thus entropy) of all closed systems increases.
It follows from equation (5) that a chemical reaction can be driven (i.e. achieve a negative ΔG) either by a suitable (i.e. negative) change in enthalpy, or by a sufficient increase in entropy, or both at the same time.
From the table values of ΔG0 or ΔG0′ we can calculate ΔG for any reaction using the equation:
| (6) |
At equilibrium, ΔG is equal to zero and the expression is equal to the equilibrium constant Keq.
Thus, equation (6) in the equilibrium state takes the form:
| (7) |
- ΔG = −R · T · ln Keq
Through this equation, we can then calculate the equilibrium constant from ΔG or vice versa.
The easy availability of ΔG0 values can lead to hasty conclusions about the impossibility of the reaction progress when finding positive ΔG0 values in the tables. But this should be avoided.
Let's show it with the following example: The metabolic pathway of glycolysis includes a step in which glucose-6-phosphate is converted to its isomer, fructose-6-phosphate. ΔG0' of this reaction is +1.7 kJ/mol. So does this mean that our cells are breaking the laws of thermodynamics by carrying out this reaction every second of our lives?
It is important to keep in mind the difference between ΔG' and ΔG0 and ΔG0. Only ΔG alone can tell us something about the thermodynamic profile of a particular reaction. The values of ΔG0 and ΔG0' are valid only when maintaining standard conditions, which are very rarely (if ever) present in nature. In order to assess the probability that a reaction will occur, we need to know the true concentrations of all the substances involved in it.
In our case, enzymes constantly remove fructose-6-phosphate by their action, so its concentration is kept low. The total ΔG of the isomerization of Glc-6-P to Fru-6-P is therefore ``negative (−2.5 kJ/mol) under the conditions found in the cell. So we see that some energetically relatively unfavorable reactions can also take place if they are combined with energetically favorable reactions (in this case, those that drain products or supply reactants).
If the transformation reaction A → B reaches an equilibrium state with a low concentration of substance B, we can combine it with the transformation reaction B → C, which, on the other hand, reaches an equilibrium state only at low concentrations of reactant B. The second reaction then effectively removes the substance B formed by the first reaction and thus prevents it from reaching a state of equilibrium.
In our example of glycolysis, the enzyme phosphofructokinase very efficiently phosphorylates Fru-6-P to fructose-1,6-bisphosphate, thereby preventing the accumulation of Fru-6-P that would inhibit the previous reaction.

![{\displaystyle \Delta G=\Delta G^{0}+R\cdot T\cdot ln{\frac {[C]^{c}\cdot [D]^{d}}{[A]^{a}\cdot [B]^{b}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/bcd22dcc82d9e6d9762126f24414f6b0c16f0d1a)
![{\displaystyle {\frac {[C]^{c}\cdot [D]^{d}}{[A]^{a}\cdot [B]^{b}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/7111a4fb16c8e7a84736b85eaad4f6f73a1ddfd5)