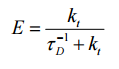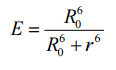Förster energy transfer
OK - BUT I WONDER WHETHER YOU UNDERSTOOD WHAT YOU WROTE
Introduction[edit | edit source]
Lots of molecules can be brought in an electronic excited state by absorbing a photon. If the emission of that photon than occurs in a singlet state, we can describe it as fluorescence. Under certain circumstances the energy of an excited molecule can be transfered to another molecule without radiation, known as förster resonance energy transfer.
The förster resonance energy transfer happens between two molecules that we can define as aceptor (A) chromophore and donator (D) chromophore. Lets assume the following: an excited (*) D chromophore molecule in the first excited singlet state (S1,0) is in a distance (r) of an A molecule (S0,0) that is in an electronic ground state. Both can not move from their position, they are embedded in a fixed matrix. The energy between the two molecular systems (D → A) is being transfered in an nonradiation process through dipole-dipole coupling:
D*(S1,0) + A(S0,0) → D(S0,0) + A*(1,0)
Depiction of the electronic excitement (kabs) of a donor molecule and its deactivations via fluorescence (kfl), inner transformation (kic) with following nonradiative deactivation (krl) and the energy transmission (kt) to an aceptor molecule.
The transmissions
D*(S1,0) → D(S0,i)
correspond to the transmissions of the excited donor molecule in the different oscillation states of the electronic ground state and reflect the fluorescent spectrum ΨD of the donator molecule.
The transmission
A(S0,0) → A*(S1,i)
corresponds to the transmission of the aceptor molecule A in the different oscillation states of the first excited S1-state and thus, corresponds to the absorption spectrum A ε of the aceptor molecule.
This emphasizes that the energy transfer is only possible, if the absorption spectrum of the acceptor with the emission spectrum of the donor overlaps. This spectral overlap is defined as J(λ).
The main part of the electronic interaction is being given by the dipole-dipole coupling. It represents the interaction of the transfer-dipole μD of the emission of the donator molecule and the transfer-dipole μA of the absorption of the aceptor molecule. The transition rate of the energy transmission can be derived by using the following equation:
k2 = relative orientation between the dipoles of the molecules QD = photon effect of the donor, if there are no acceptor molecules n = refraction index NA = avogadro constant r = distance between donor and acceptor τ = fluorescending life span of the donor, if there are no aceptor molecules R0 = Förster-distance εA = molar decadic absorption coefficient of the aceptor ΨDnorm = standardized fluorescent spectrum of the donator:
If we compare these equations we will get R0:
The transmission of energy is in concurrence with the fluorescending process of the donor molecule. Short transfer-times result in an effective energy transfer to the aceptor and vice versa. The energy transfer efficiency E is defined as the part of the absorpt photons that the donator transfers to the aceptor:
We can put this equation in the one mentioned above:
this equals to:
The graph of the energy transfer efficiency of the donor-aceptor-radius shows a functional connection:
If r=R0 the transfer efficiency is 50% (definition of the Förster radius).
Discussion[edit | edit source]
The strong distance-dependence of the förster resonance energy transfer efficiency has been widely utilized in studying the structure and dynamics of proteins and nucleic acids, in the detection and visualization of intermolecular association and in the development of intermolecular binding assays. Förster resonance energy transfer is a particularly useful tool in molecular biology as the fraction, or efficiency, of energy that is transferred can be measured, and depends on the distance between the two fluorophores.
References[edit | edit source]
Methoden der Biophysikalischen Chemie; 2. Edition; Roland Winter, Frank Noll, Claus Czeslik Kurzlehrbuch Physikalische Chemie; 4. Edition; P. W. Atkins, Julio de Paula http://onlinelibrary.wiley.com/doi/10.1002/andp.19484370105/abstract: Förster T: Zwischenmolekulare Energiewanderung und Fluoreszenz. In: Ann. Physik. 437, 1948, S. 55 Goddard JP, Reymond JL: Recent advances in enzyme assays. In: Trends Biotechnol.. 22, Nr. 7, Juli 2004, S. 363–370 Clegg, Robert M.: The history of FRET. In: Lakowicz, Joseph R.; Chris D. Geddes (Hrsg.): Reviews in Fluorescence 2006. Springer, Berlin 2006 Marullo S, Bouvier M: Resonance energy transfer approaches in molecular pharmacology and beyond, August 2007, S. 362–365