Electron shell of the atom
It may contain one or more electrons.
Electron[edit | edit source]
A microparticle and carrier of negative charge, denoted by e or e−. Electrons in an atom exist only in states with specific energy.
Electrons can emit or absorb energy only in discrete quanta, during transitions from one energy level to another. The energy of a quantum is given by the difference between the energies corresponding to specific energy levels in the atomic shell.
The electron has a dual character — wave and particle. This is also reflected in the uncertainty principle (it is not possible to determine both the exact position and velocity of an electron). We can only determine probability — the region with the highest probability of finding an electron is called an orbital.
Quantum numbers[edit | edit source]
The state of an electron in the atomic shell is described by four quantum numbers (three characterize the orbital, and the fourth describes the projection of the electron’s spin in the orbital).
- Principal quantum number (n) – expresses the energy of the orbital. It takes values from 1 to 7 (also denoted as K, L, M, N, O, P, Q).
- Azimuthal (orbital) quantum number (l) – takes values from 0 to n−1. It determines the spatial shape and energy differences of orbitals.
- Magnetic quantum number (m) – determines the orientation of the orbital in a magnetic field. It takes values from −l to +l.
- Spin quantum number (s) – takes values +1/2 or −1/2. A maximum of two electrons can occupy one orbital, and they must have opposite spins, forming an electron pair.
| Electron shell | n | l | Orbital type | m values | Number of orbitals | Max. number of electrons |
|---|---|---|---|---|---|---|
| K | 1 | 0 | 1s | 0 | 1 | 2 |
| L | 2 | 0
1 |
2s
2p |
0
−1, 0, +1 |
1
3 |
8 |
| M | 3 | 0
1 2 |
3s
3p 3d |
0
−1, 0, +1 −2, −1, 0, +1, +2 |
1
3 5 |
18 |
| N | 4 | 0
1 2 3 |
4s
4p 4d 4f |
0
−1, 0, +1 −2, −1, 0, +1, +2 −3, −2, −1, 0, +1, +2, +3 |
1
3 5 7 |
32 |
Rules for filling electron orbitals[edit | edit source]
Electron configuration (of ions) – the arrangement of electrons in individual orbitals within the electron shells. From this, it is possible to determine the ability of an atom (ion) to form certain types of chemical bonds. The order of filling is determined by the Aufbau principle, the Pauli exclusion principle, and Hund’s rule.
Aufbau principle – the principle of minimum energy: an atom in its ground state tends to occupy the lowest possible energy state. Orbitals are filled starting from those with the lowest energy:
1s→2s→2p→3s→3p→4s→3d→4p→5s→4d→5p→6s→4f→5d→6p→7s→5f→6d→7p
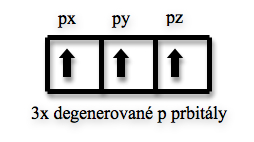
Degenerate orbitals – orbitals that have the same energy (they have the same principal and azimuthal quantum numbers but differ in the magnetic quantum number).
Pauli exclusion principle – in an atom, there are no two electrons that have all four quantum numbers identical (they must differ at least in spin, which is represented by opposite arrows).
Hund’s rule – Degenerate orbitals are filled in such a way that the number of unpaired electrons is maximized (with parallel spins) before electron pairs begin to form.