Atomic Models
|
Check of this article is requested. Suggested reviewer: RNDr. Petr Heřman |
Atomic models[edit | edit source]
There has been a variety of atomic models throughout history of atomic physics, that refers mainly to a period from the beginning of 19th century to the first half of 20th century, when a final model of atom which is being used nowadays (or accepted as the most accurate one) was invented. Although the awareness of atom existence goes way back to the antique period of the world history (Greek conception of atom), this article will be mainly about five basic atomic models, from which each one has somehow contributed to how we percept the structure of atom itself - Dalton´s Billiard Ball Model, J.J Thomson's "plum pudding" model, Rutherford's Planetary model, Bohr's Atomic model, Electron Cloud Model/Quantum Mechanics Model.
John Dalton’s atomic model[edit | edit source]
John Dalton was an English scientist, who came up with an idea that all matter is composed of very small things. It was the first complete attempt to describe all matter in terms of particles. He called these particles atoms and formed an atomic theory. In this theory he claims that:
- All matter is made of atoms. Atoms are indivisible and indestructible
- All atoms of a given element are identical in mass and properties
- Compounds are formed by a combination of two or more different kinds of atoms
- A chemical reaction is a rearrangement of atoms
Parts of his theory had to be modified based on the discovery of subatomic particles and isotopes. We now also know that atoms are not indivisible, because they are made up of neutrons, electrons and protons.
Plum pudding model[edit | edit source]
After discovery of an electron in 1897, people realised that atoms are made up of even smaller particles. Shortly after in 1904 J. J. Thomson proposed his famous “plum pudding model“. In this model, atoms were known to consist of negatively charged electrons, however the atomic nucleus had not been discovered yet. Thomson knew that atom had an overall neutral charge. He thought that there must be something to counterbalance the negative charge of an electron. He came up with an idea that negative particles are floating within a soup of diffuse positive charge. His model is often called the plum pudding model, because of his similarity to a popular English dessert.
Rutherford’s model of the atom[edit | edit source]
Rutherford was first, who suggested that Thomson’s plum pudding model was incorrect. His new model introduces nucleus to the atom theory. Nucleus contains relatively high central charge concentrated into very small volume. This small volume also contains the bulk of the atomic mass of the atom. Nucleus is surrounded by lighter and negatively charged electrons. His model is sometimes known as the planetary model of the atom. However, there were still some major problems with this model. For example Rutherford could not explain why atoms only emit light at certain frequencies. This problem was solved later by a Danish physicist Niels Henrik David Bohr.
Bohr’s model of the atom[edit | edit source]
Bohr model describes the atom as a positively charged nucleus, which is surrounded by electrons. Electrons travel in circular orbits, attraction is provided by electrostatic forces. Normally occupied energy level of the electron is called the ground state. The electron can move to the less – stable level by absorbing energy. This higher – energy level is called excited state. The electron can return to its original level by releasing the energy. All in all, when electron jumps between orbits, it is accompanied by an emitted or absorbed amount of energy (hv).
Electron Cloud Model/Quantum Mechanics Model of Atom[edit | edit source]
Quantum Mechanics Model of Atom is nowadays being taught as the most "realistic" atomic model that describes atomic mechanisms as how present science presumes they work. It came to exist as a result of combination of number of scientific assumptions:
- All particles could be percieved as matter waves with a wavelength. (Louis de Broglie)
- Resulting from the previous assumption, atomic model which treats electrons also as matter waves was proposed. (Erwin Schrödinger, quantum mechanical atomic model emerged from the solution of Schrödinger's equation for electron in central electrical field of nucleus.)
- Principle of uncertainty states that we can't know both the energy and position of an electron. Therefore, as we learn more about the electron's position, we know less about its energy, and vice versa. (Werner Heisenberg)
- There exists more than one energy level of electron in the atom. Electrons are assigned certain atomic orbitals, that can differ from one another in energy. (Niels Bohr)
- Electrons have an intrinsic property called spin, and an electron can have one of two possible spin values: spin-up or spin-down. Any two electrons occupying the same orbital must have opposite spins. (the Stern-Gerlach Experiment)
Basic description of the quantum mechanical atomic model:[edit | edit source]
Quantum mechanics physics propose that electrons are moving around the nucleus not on specifically defined electron paths (as we have seen eg. in Rutherford's planetary atomic model), but in a certain three dimensional space (atomic orbital), in which their own occurrence has a certain probability, meaning their position cannot be calculated with 100% accuracy.
Four numbers, called quantum numbers, were introduced to describe the characteristics of electrons and their orbitals.
Quantum numbers[edit | edit source]
Principal quantum number: n[edit | edit source]
- describes the energy level of the electron in an atom (it also describes the average distance of the orbital from nucleus)
- It has positive whole number values: 1, 2, 3, 4,... (theoretically speaking the numbers could go to infinite, practically there are 7 known energy levels), it can be seen sometimes described in capital letters instead of numbers, beginning with K (K, L, M, N...)
- the n value describes the size of the orbital
Angular momentum quantum number: l[edit | edit source]
- describes basically the shape of the orbital
- This number is limited by the principal quantum number. Its value goes from 0 to n-1. For example, for orbitals with principal quantum number n=2 there can by 2 different shapes of orbitals (2 different values l=0 and l=1)
- for every number exists a letter describing the shape of the orbital as shown in the table below
- Value of l (subshell) Letter
| Value of l (subshell) | Letter |
|---|---|
| 0 | s |
| 1 | p |
| 2 | d |
| 3 | f |
| 4 | g |
Magnetic quantum number: ml[edit | edit source]
- describes how different shapes of orbitals are oriented in space. Its value can be from -l to 0 to +l. For example, for value l=1 there exists 3 values m= -1, 0, +1, meaning that the shape of that orbital can be oriented in 3 different ways in space.
| Value of l | Values of ml |
|---|---|
| 0 | 0 |
| 1 | -1,0,+1 |
| 3 | -3,-2,-1,0,+1,+2,+3 |
Spin quantum number: ms[edit | edit source]
- describes in which direction is an electron spinning in a magnetic field . That can be either clockwise or counterclockwise and as a result, there are only 2 values allowed: -1/2 and +1/2.
- One consequence of electron spin is that a maximum of two electrons can occupy any given orbital, and the two electrons occupying the same orbital must have opposite spin. This is also called the Pauli exclusion principle.
Principles of atom structure[edit | edit source]
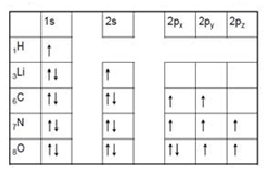
Based on knowledge of quantum numbers, we are now able to build an electron configuration of an atom that describes electron arrangement in an atom. Apart from Pauli exclusion principle, there are 2 other rules that we must follow:
- Aufbau principle: Each electron occupies the lowest energy orbital available.
- Hund's rule: a single electron with the same spin must occupy each orbital in a sublevel before they pair up with an electron with an opposite spin.
The importance of quantum mechanical atomic model has 2 main aspects. First, being able to build an electron structure of atoms of specific substances helps us to understand how the atoms interact in molecules, therefore we are one step closer to a more detailed description of attributes of those substances. Second, it leaves an open door to more potential theories which could expand our knowledge and perception of the world and universe surrounding us.
Sources:[edit | edit source]
- What are the 6 models of the atom? | Socratic. (2015, May 23). Retrieved October 29, 2017, from https://socratic.org/questions/what-are-the-6-models-of-the-atom
- Kvantově mechanický model atomu. (n.d.). Retrieved October 29, 2017, from http://www.dobreznamky.cz/kvantove-mechanicky-model-atomu/
- Stavba atomového obalu. (2008, October 4). Retrieved October 29, 2017, from https://www.odmaturuj.cz/fyzika/stavba-atomoveho-obalu/
- The quantum mechanical model of the atom. (n.d.). Retrieved October 29, 2017, from https://www.khanacademy.org/science/physics/quantum-physics/quantum-numbers-and-orbitals/a/the-quantum-mechanical-model-of-the-atom
- Benešová, M., & Satrapová, H. (2002). Odmaturuj z chemie. Didaktis.
- Plum pudding model. (2017, October 12). Retrieved October 29, 2017, from https://en.wikipedia.org/wiki/Plum_pudding_model
- Bohr model. (2017, October 19). Retrieved October 29, 2017, from https://en.wikipedia.org/wiki/Bohr_model
- The Editors of Encyclopædia Britannica. (2014, June 05). Bohr atomic model. Retrieved October 29, 2017, from https://www.britannica.com/science/Bohr-atomic-model
- Orbital shapes and their orientation for different angular momentum and magnetic numbers - MEFANET, síť lékařských fakult ČR a SR. (n.d.). Orbital. Retrieved October 29, 2017, from http://www.wikiskripta.eu/w/Orbital#/media/File:Single_electron_orbitals.jpg
- Ilustration of Thomson's perception of atom - W. (n.d.). Retrieved October 29, 2017, from https://www.meritnation.com/ask-answer/question/why-is-thomsons-model-also-known-as-plum-pudding-model/structure-of-the-atom/1299876
- Basic ilustration of the Aufbau principle - Electronic configuration. (n.d.). Retrieved October 29, 2017, from http://www.chemie.utb.cz/rvicha/Sac/vystprincip.html
- Hund's Rule of Maximum Spin Multiplicity. (n.d.). Retrieved October 29, 2017, from http://www.eurekasparks.org/2015/07/hunds-rule-of-maximum-spin-multiplicity.html
- Examples of atoms described by quantum numbers - (n.d.). Retrieved October 29, 2017, from http://chemie-obecna.blogspot.cz/2011/08/radioaktivita.html