Polarimetry (2. LF UK)
(Redirected from Polarimetry Practical)
|
Check of this article is requested. Suggested reviewer: Carmeljcaruana |
Introduction[edit | edit source]
Definition[edit | edit source]
Polarimetry is an optical method used to measure chemical concentration in solution.
Principle[edit | edit source]
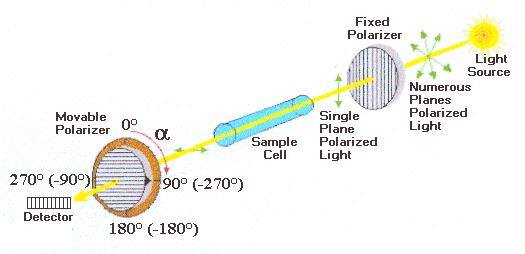
Light waves are electromagnetic (EM) waves. EM waves consist of perpendicular oscillating electric and magnetic fields; the direction of oscillation of both fields is random although they are at all times perpendicular to each other. Light waves which only oscillate in one direction are called polarized. Light can be polarised by passing it through a polarizing filter. Some substances can rotate the the polarisation of light ('optically active substances'), the higher the concentration and the higher the length of solution through which the light passes the larger the angle of rotation. Polarimeters consist of a sodium lamp which emits monochromatic yellow light which is passed through a polarizer. If this polarised light is passed through an optically active substance its plane of polarisation will be rotated, my measuring the angle of rotation one can calculate the concentration. The specific optical rotation [α] is the rotation per unit distance of cuvette and per unit concentration.
Importance In Clinical Medicine[edit | edit source]
The specific rotation is often used in pharmaceutical industry for the identification and purity control of chiral substances (which are always optically active). Of particular importance is the measurement of concentrations of amino acids, terpenes and sugars since the majority of these substances are optically active.
Literature review [edit | edit source]
Advantages and disadvantages[edit | edit source]
For the polarimeter large sample volumes with high substance concentration are required. However, the sample used remains unchanged after measurement. Due to the low equipment complexity, the measurement is very simple and fast, which means that costs are low. In addition polarimetry is quite specific, because only a few substances rotate polarized light. However, the result may not be very accurate because the method involves the subjective assessment of brightness of light by the user.
How does it work?[edit | edit source]
Plane-polarized light is introduced to a tube containing a solution with the substance to be measured. If the substance is optical inactive, the plane of the polarized light will not change in orientation and the observer will read an angle of [α]= 0. If the compound is optical active, the plane of the light would be rotated. In order to observe the maximum brightness, the observer (person or instrument) will have to rotate the axis of the analyzer back, either clockwise or counterclockwise direction depending on the nature of the compound. For clockwise direction, the rotation (in degrees) is defined as positive ("+") and called dextrorotatory (from the Latin: dexter=right). In contrast, the counterclockwise direction is defined as negative ("-") and called levorotatory (from the Latin laevus=left). The observed rotation α depends on the specific rotation of the substance [α], the length of the tube and the concentration of the optical active compound (and frequency of light and temperature).
Risks[edit | edit source]
There are no major risks associated with polarimetry, neither for the patient nor for the operator.
Ethical issues[edit | edit source]
Polarimetry does not raise any ethical issues
Construction of the Instrument[edit | edit source]
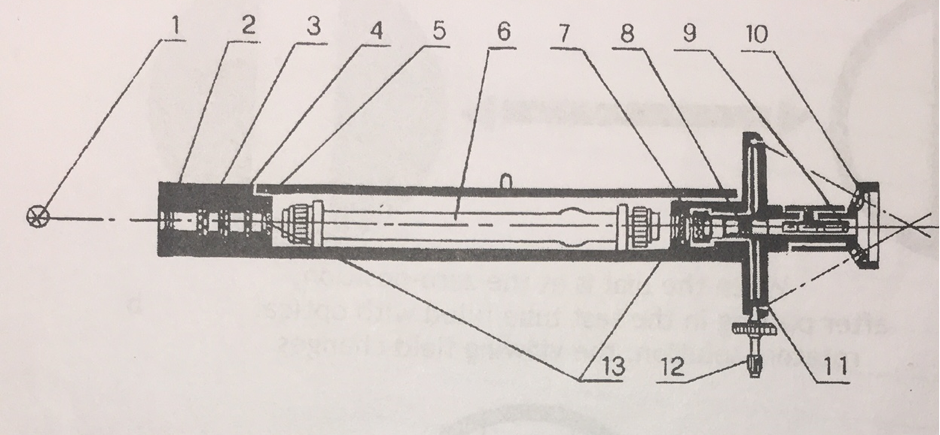
System diagram of the instrument
1.) Light Source (Sodium Light) 2.) Collector Lens 3.) Colour Filter 4.) Polarizer 5.) Half-wave Plate 6.) Test Tube with solution 7.) Polarization Analyzer 8.) Object Lens 9.) Eye Lens 10.) Magnifying Glass 11.) Dial Vernier 12.) Dial Rotary Hand-wheel 13.) Protective Plate
Manual Polarimeter; lateral view

Manual Polarimeter; superior view

Manual Polarimeter; superior view with testtube

Methodology[edit | edit source]

1th Task[edit | edit source]
It is necessary to standardize the measurement before measuring the specific rotation and determining the unknown glucose concentration. This is the so-called zero point correction and the reasons for this correction are incorrect alignment and contamination of the optical parts.
- Use a 10cm long cuvette with distilled water marked DW (distilled water) for this measurement. You must catch any bubbles in the bulge of the cuvette when inserting the cuvette. Never touch the ends of the cuvette to avoid contamination.
- Insert the cuvette with distilled water into the cuvette compartment and set the vernier scale to a value out of 0° (e.g. 10°). Use two pies of magnifying glasses located (right and left) on the eyepiece cups when reading values on the vernier scale.
- Focus the eyepiece (the rotating ring on the eyepiece) so that two sharp cutting edges between the three light fields are visible in the eyepiece.
- Then turn the analyzer towards 0° until the cutting edges between the three light fields disappears in the eyepiece and the intensity is the equal for each light field.
- Read the value on the vernier scale and write it in the protocol.
- Repeat the measurement five times and write the value on the vernier scale in the protocol (each team member should take turns). Be sure to rotate the analyzer before each measurement so that the equal intensity of light fields disappears in the eyepiece and individual cutting edge appear between them (each team member should adjust the focus of the eyepiece to suit the sharpness of the cutting edges).
- Calculate the average and standard deviation.
2nd Task[edit | edit source]
Determine the specific rotation of glucose by measuring the rotation of a glucose solution of known concentration. Compare the result with the table values. Table value of specific glucose rotation Failed to parse (syntax error): {\displaystyle [\alpha]_D^{20°C}} = 52,5°.
- Use a 20cm long cuvette in which there is a solution of glucose in distilled water with a concentration of 10g/100ml to measure the specific rotation, .
- Insert a cuvette with a solution of known concentration into the cuvette compartment and set the vernier scale to 0°.
- Focus the eyepiece so that two sharp cutting edges between the three light fields are visible in the eyepiece.
- Then rotate the analyzer until the cutting edges between the three light fields disappears in the eyepiece and the intensity is the equal for each light field.
- Repeat the measurement ten times similarly to standardization and write the value on the vernier scale in the protocol.
- Calculate the average and standard deviation.
- Calculate the optical rotation correction obtained by subtracting the average rotation value when standardizing the measurement from the average optical rotation value of a sample with a known glucose concentration.
- Calculate the specific rotation of the glucose and report its value and the formula you used.
- Compare the result with the table values.
Recommended relation
,
where c is a concentration in g/100ml, α is a measured angle of rotation, l is a length of cuvette and [α] is a specific optical rotation.
3rd Task[edit | edit source]
Based on the optical rotation measurement, determine the concentration of the optically active substance (glucose) in the solution using the specific rotation calculated in the previous step.
- Use a cuvette 10cm long in which there is a solution of a glucose sample in distilled water of unknown concentration to measure the unknown concentration.
- Insert a cuvette with a solution of unknown concentration into the cuvette compartment and set the vernier scale back to 0°.
- Focus the eyepiece so that two sharp cutting edges between the three light fields are visible in the eyepiece.
- Then rotate the analyzer until the cutting edges between the three light fields disappears in the eyepiece and the intensity is the equal for each light field.
- Repeat the measurement ten times as in the previous two tasks and write the value on the vernier scale in the protocol.
- Calculate the average and standard deviation.
- Calculate the optical rotation correction obtained by subtracting the average rotation value when standardizing the measurement from the average optical rotation value of a sample with an unknown glucose concentration.
- Calculate the glucose concentration using the measured specific rotation in the previous task and report its value and the formula you used.
Recommended relation
,
where c is a concentration in g/100ml, α is a measured angle of rotation, l is a length of cuvette and [α] is a specific optical rotation.
Future of Polarimetry[edit | edit source]
The future plans of polarimetry are to make polarimeters smaller, cheaper, mobile, simpler and more precise. Every year new advanced models are developed. Scientists are claiming that in the near future a microchip sized polarimeter will be invented, which will be a major development.
Reference list[edit | edit source]
1.) Rudolph Research Analytical; http://rudolphresearch.com/products/polarimeters/polarimetry-definitions/ 2.) Rudolph Research Analytical; http://www.chem.ucla.edu/~bacher/General/30BL/tips/Polarimetry.html 3.) ChemgaPedia; http://www.chemgapedia.de/vsengine/about/de/index.html 4.) Fun Man FUNG (Video); https://www.youtube.com/watch?v=T6zjiU_-91g 5.) TheSimpleChemics (Video); https://www.youtube.com/watch?v=l_G34WeJjgs&t=36s 6.) Andreas Jerrentrup; 1.ÄP Physik für Mediziner; 17th edition; 2006; ISBN: 978-3-13-114937-4







![{\displaystyle \alpha ={\cfrac {[\alpha ]\cdot l\cdot c}{100}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/c441430795cf7ef914543ad4913db271cca38a8d)