Albumin
Albumin is a serum protein that accounts for 55–65% of total serum protein (the average plasma albumin concentration is 40 g/l[1]). It is synthesized in the liver and its formation depends on the intake of amino acids.
Albumin:
- Albumin plays a significant role in maintaining the oncotic pressure of the plsama. Albumin values lower than 20 g/l are associated with the occurence of edema.
- Performs a transport functions. It transports bilirubin, heme, steroid substances, thyroxine, fatty acids, bile acids, metals, drugs and other substances.
- It creates the body's protein reserve and serves as a source of amino acids, especially essential amino acids for various tissues. Its concentration decreases with malnutrition. However, serum albumin concentration is not a good indicator of early protein malnutrition, because in amino acid deficiency, albumin catabolism is also reduced and albumin is translocated from the interstitium to maintain adequate plasma levels. The slow decline is also due to a longer half-life and a large body supply. For this reason, it does not reflect the full extent of the nutritional deficit.
Albumin synthesis[edit | edit source]
Albumin synthesis begins in the nucleus of hepatocytes by transcription of genes into mRNA, which is then secreted into the cytoplasm, where it binds to ribosomes to form polysomes that form preproalbumin. Preproalbumin enters the endoplasmic reticulum where it is converted to proalbumin, the major intracellular form of albumin. Proalbumin is further secreted into the Golgi apparatus, where it is transformed into albumin and immediately excreted from the liver.[2]
Determination of albumin[edit | edit source]
__Deteremining the concentration of albumin in the serum is one of the widespread biochemical tests, especially suitable for liver and kidney diseases and assessment of nutritional status. A decrease in albumin can accompany chronic inflammation and an increase in catabolism in some disease states. Other causes of decreased or increased albumin concentration are similar to those of total proteins.
The principle of albumin determination[edit | edit source]
In a weakly acidic environment, albumin behaves as a cation. It can react with anionic dyes and an albumin-dye complex is formed. Binding of the dye is accompanied by a change in its color. Bonding occurs in a slightly acidic environment in the presence of surfactants.
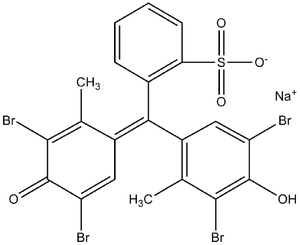
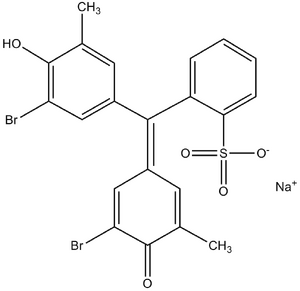
Organic dyes are used, which react with albumin much faster than with other serum proteins. They most often contain the group -SO3H. An example can be bromcresol purple or bromcresol green.
Reference range: Serum albumin concentration (S-albumin): 35–53 g/l
More sensitive immunochemical methods (most often immunoturbidimetry) are usually used to determine albumin in other fluids, e.g. urine or cerebrospinal fluid.
Links[edit | edit source]
Related articles[edit | edit source]
Reference[edit | edit source]
- ↑ ŠVÍGLEROVÁ, Jitka. Albumin [online]. The last revision 2009-02-18, [cit. 2010-10]. <https://web.archive.org/web/20160416224413/http://wiki.lfp-studium.cz/index.php/Albumin>.
- ↑ PERALTA, R. Hypoalbuminemia : Pathophysiology [online]. Medscape, ©2015. The last revision -, [cit. 2016-02-03]. <https://emedicine.medscape.com/article/166724-overview>.